The role of VEGF 165b in pathophysiology
- PMID: 23076130
- PMCID: PMC3547904
- DOI: 10.4161/cam.22439
The role of VEGF 165b in pathophysiology
Abstract
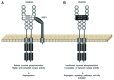
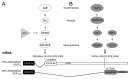
Anti-angiogenic vascular endothelial growth factor A (VEGF) 165b and pro-angiogenic VEGF 165 are generated from the same transcript, and their relative amounts are dependent on alternative splicing. The role of VEGF 165b has not been investigated in as much detail as VEGF 165, although it appears to be highly expressed in non-angiogenic tissues and, in contrast with VEGF 165, is downregulated in tumors and other pathologies associated with abnormal neovascularization such as diabetic retinopathy or Denys Drash syndrome. VEGF 165b inhibits VEGFR2 signaling by inducing differential phosphorylation, and it can be used to block angiogenesis in in vivo models of tumorigenesis and angiogenesis-related eye disease. Recent reports have identified three serine/arginine-rich proteins, SRSF1, SRSF2 and SRSF6, and studied their role in regulating terminal splice-site selection. Since the balance of VEGF isoforms is lost in cancer and angiogenesis-related conditions, control of VEGF splicing could also be used as a basis for therapy in these diseases.
Figures

Similar articles
-
The anti-angiogenic isoforms of VEGF in health and disease.Biochem Soc Trans. 2009 Dec;37(Pt 6):1207-13. doi: 10.1042/BST0371207. Biochem Soc Trans. 2009. PMID: 19909248 Free PMC article.
-
Overexpression of VEGF165b, an inhibitory splice variant of vascular endothelial growth factor, leads to insufficient angiogenesis in patients with systemic sclerosis.Circ Res. 2011 Jul 22;109(3):e14-26. doi: 10.1161/CIRCRESAHA.111.242057. Epub 2011 Jun 2. Circ Res. 2011. PMID: 21636803
-
WT1 mutants reveal SRPK1 to be a downstream angiogenesis target by altering VEGF splicing.Cancer Cell. 2011 Dec 13;20(6):768-80. doi: 10.1016/j.ccr.2011.10.016. Cancer Cell. 2011. PMID: 22172722 Free PMC article.
-
[Alternative splicing of vascular endothelial growth factor A and ocular neovascularization].Zhonghua Yan Ke Za Zhi. 2011 Apr;47(4):373-7. Zhonghua Yan Ke Za Zhi. 2011. PMID: 21612688 Review. Chinese.
-
How VEGF-A and its splice variants affect breast cancer development - clinical implications.Cell Oncol (Dordr). 2022 Apr;45(2):227-239. doi: 10.1007/s13402-022-00665-w. Epub 2022 Mar 18. Cell Oncol (Dordr). 2022. PMID: 35303290 Free PMC article. Review.
Cited by
-
Shift in VEGFA isoform balance towards more angiogenic variants is associated with tumor stage and differentiation of human hepatocellular carcinoma.PeerJ. 2018 Jun 5;6:e4915. doi: 10.7717/peerj.4915. eCollection 2018. PeerJ. 2018. PMID: 29888133 Free PMC article.
-
Angiogenesis and hypoxia in the kidney.Nat Rev Nephrol. 2013 Apr;9(4):211-22. doi: 10.1038/nrneph.2013.35. Epub 2013 Mar 5. Nat Rev Nephrol. 2013. PMID: 23458926 Review.
-
Macrophage IL-1β promotes arteriogenesis by autocrine STAT3- and NF-κB-mediated transcription of pro-angiogenic VEGF-A.Cell Rep. 2022 Feb 1;38(5):110309. doi: 10.1016/j.celrep.2022.110309. Cell Rep. 2022. PMID: 35108537 Free PMC article.
-
Association between the ratio of anti-angiogenic isoform of VEGF-A to total VEGF-A and adverse clinical outcomes in patients after acute myocardial infarction.Int J Cardiol Heart Vasc. 2018 Mar 22;19:3-7. doi: 10.1016/j.ijcha.2018.03.004. eCollection 2018 Jun. Int J Cardiol Heart Vasc. 2018. PMID: 29946556 Free PMC article.
-
Antiangiogenic VEGF165b Regulates Macrophage Polarization via S100A8/S100A9 in Peripheral Artery Disease.Circulation. 2019 Jan 8;139(2):226-242. doi: 10.1161/CIRCULATIONAHA.118.034165. Circulation. 2019. PMID: 30586702 Free PMC article.
References
-
- Bates DO, Cui TG, Doughty JM, Winkler M, Sugiono M, Shields JD, et al. VEGF165b, an inhibitory splice variant of vascular endothelial growth factor, is down-regulated in renal cell carcinoma. Cancer Res. 2002;62:4123–31. - PubMed
-
- Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z. Vascular endothelial growth factor (VEGF) and its receptors. FASEB J. 1999;13:9–22. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
