Complex intramolecular mechanics of G-actin--an elastic network study
- PMID: 23077498
- PMCID: PMC3471905
- DOI: 10.1371/journal.pone.0045859
Complex intramolecular mechanics of G-actin--an elastic network study
Abstract
Systematic numerical investigations of conformational motions in single actin molecules were performed by employing a simple elastic-network (EN) model of this protein. Similar to previous investigations for myosin, we found that G-actin essentially behaves as a strain sensor, responding by well-defined domain motions to mechanical perturbations. Several sensitive residues within the nucleotide-binding pocket (NBP) could be identified, such that the perturbation of any of them can induce characteristic flattening of actin molecules and closing of the cleft between their two mobile domains. Extending the EN model by introduction of a set of breakable links which become effective only when two domains approach one another, it was observed that G-actin can possess a metastable state corresponding to a closed conformation and that a transition to this state can be induced by appropriate perturbations in the NBP region. The ligands were roughly modeled as a single particle (ADP) or a dimer (ATP), which were placed inside the NBP and connected by elastic links to the neighbors. Our approximate analysis suggests that, when ATP is present, it stabilizes the closed conformation of actin. This may play an important role in the explanation why, in the presence of ATP, the polymerization process is highly accelerated.
Conflict of interest statement
Figures
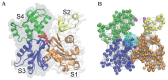


 (grey bead), connected by an elastic link. The ADP is elastically connected to its neighbors and the phosphate is elastically linked to the three sensitive nodes (red beads).
(grey bead), connected by an elastic link. The ADP is elastically connected to its neighbors and the phosphate is elastically linked to the three sensitive nodes (red beads).
 is visualized as a small red bead; ATP is modeled as a dimer consisting of ADP and Pi. Magenta-colored beads indicate residues between which additional breakable links can become established. Such breakable links are shown by solid magenta lines, if they are actually present, and by dashed lines if they are broken.
is visualized as a small red bead; ATP is modeled as a dimer consisting of ADP and Pi. Magenta-colored beads indicate residues between which additional breakable links can become established. Such breakable links are shown by solid magenta lines, if they are actually present, and by dashed lines if they are broken.
Similar articles
-
Nucleotide-dependence of G-actin conformation from multiple molecular dynamics simulations and observation of a putatively polymerization-competent superclosed state.Proteins. 2009 Aug 1;76(2):353-64. doi: 10.1002/prot.22350. Proteins. 2009. PMID: 19156817
-
A nucleotide state-sensing region on actin.J Biol Chem. 2010 Aug 13;285(33):25591-601. doi: 10.1074/jbc.M110.123869. Epub 2010 Jun 8. J Biol Chem. 2010. PMID: 20530485 Free PMC article.
-
Switch II mutants reveal coupling between the nucleotide- and actin-binding regions in myosin V.Biophys J. 2012 Jun 6;102(11):2545-55. doi: 10.1016/j.bpj.2012.04.025. Epub 2012 Jun 5. Biophys J. 2012. PMID: 22713570 Free PMC article.
-
Actin polymerization: regulation by divalent metal ion and nucleotide binding, ATP hydrolysis and binding of myosin.Adv Exp Med Biol. 1994;358:71-81. doi: 10.1007/978-1-4615-2578-3_7. Adv Exp Med Biol. 1994. PMID: 7801813 Review.
-
The complexity and diversity of the actin cytoskeleton of trypanosomatids.Mol Biochem Parasitol. 2020 May;237:111278. doi: 10.1016/j.molbiopara.2020.111278. Epub 2020 Apr 28. Mol Biochem Parasitol. 2020. PMID: 32353561 Review.
Cited by
-
Fractal globules: a new approach to artificial molecular machines.Biophys J. 2014 Nov 18;107(10):2361-8. doi: 10.1016/j.bpj.2014.10.019. Biophys J. 2014. PMID: 25418305 Free PMC article.
-
Structure-Encoded Global Motions and Their Role in Mediating Protein-Substrate Interactions.Biophys J. 2015 Sep 15;109(6):1101-9. doi: 10.1016/j.bpj.2015.06.004. Epub 2015 Jul 2. Biophys J. 2015. PMID: 26143655 Free PMC article. Review.
-
Coarse-Grained Protein Dynamics Studies Using Elastic Network Models.Int J Mol Sci. 2018 Dec 5;19(12):3899. doi: 10.3390/ijms19123899. Int J Mol Sci. 2018. PMID: 30563146 Free PMC article. Review.
-
Simple mechanics of protein machines.J R Soc Interface. 2019 Jun 28;16(155):20190244. doi: 10.1098/rsif.2019.0244. Epub 2019 Jun 19. J R Soc Interface. 2019. PMID: 31213170 Free PMC article.
-
Computational Ways to Enhance Protein Inhibitor Design.Front Mol Biosci. 2021 Feb 3;7:607323. doi: 10.3389/fmolb.2020.607323. eCollection 2020. Front Mol Biosci. 2021. PMID: 33614705 Free PMC article.
References
-
- Alberts B, Johnson A, Lewis J, Raff M, Roberts K, et al... (2008) Molecular Biology of the Cell. Garland Press, 5th edition.
-
- Korn ED, Carlier MF, Pantaloni D (1987) Actin polymerization and ATP hydrolysis. Science 238: 638–644. - PubMed
-
- Otterbein LR, Graceffa P, Dominguez R (2001) The crystal structure of uncomplexed actin in the ADP state. Science 293: 708–11. - PubMed
-
- Graceffa P, Dominguez R (2003) Crystal structure of monomeric actin in the ATP state. Structural basis of nucleotide-dependent actin dynamics. The Journal of Biological Chemistry 278: 34172–80. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

