New concepts of endoplasmic reticulum function in the heart: programmed to conserve
- PMID: 23085588
- PMCID: PMC3557761
- DOI: 10.1016/j.yjmcc.2012.10.006
New concepts of endoplasmic reticulum function in the heart: programmed to conserve
Abstract
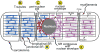
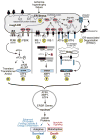
Secreted and membrane proteins play critical roles in myocardial health and disease. Studies in non-myocytes have shown that the peri-nuclear ER is the site for synthesis, folding, and quality control of most secreted and membrane proteins, as well as a nexus of a signal transduction system, called the ER stress response, which informs the cell about the status of ER protein folding. Moreover, the dynamic physical and functional association of the ER with mitochondria is a key site responsible for integrating ER function and mitochondrial metabolism, but is only just beginning to be understood in the myocardium. Although a great deal is known about roles played by the sarcoplasmic reticulum (SR) in contractile calcium handling in the heart, little is known about the relative locations and functions of the peri-nuclear ER and the SR in terms of secreted and membrane protein synthesis and folding. In this review we will explore the current state of knowledge of the location of secreted and membrane protein synthesis, folding, and quality control machinery in cardiac myocytes, as well as our understanding of the functional consequences of ER stress and the unfolded protein response in the heart in terms of protein synthesis, cell growth, and metabolic regulation. This article is part of a Special Issue entitled "Focus on Cardiac Metabolism".
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures
Similar articles
-
Roles for ATF6 and the sarco/endoplasmic reticulum protein quality control system in the heart.J Mol Cell Cardiol. 2014 Jun;71:11-5. doi: 10.1016/j.yjmcc.2013.09.018. Epub 2013 Oct 16. J Mol Cell Cardiol. 2014. PMID: 24140798 Free PMC article. Review.
-
Biology of endoplasmic reticulum stress in the heart.Circ Res. 2010 Nov 12;107(10):1185-97. doi: 10.1161/CIRCRESAHA.110.227033. Circ Res. 2010. PMID: 21071716 Review.
-
Roles for the sarco-/endoplasmic reticulum in cardiac myocyte contraction, protein synthesis, and protein quality control.Physiology (Bethesda). 2012 Dec;27(6):343-50. doi: 10.1152/physiol.00034.2012. Physiology (Bethesda). 2012. PMID: 23223628 Review.
-
Endoplasmic reticulum stress and diabetic cardiomyopathy.Exp Diabetes Res. 2012;2012:827971. doi: 10.1155/2012/827971. Epub 2011 Nov 24. Exp Diabetes Res. 2012. PMID: 22144992 Free PMC article. Review.
-
Endoplasmic reticulum proteins in cardiac development and dysfunction.Can J Physiol Pharmacol. 2009 Jun;87(6):419-25. doi: 10.1139/y09-032. Can J Physiol Pharmacol. 2009. PMID: 19526035 Review.
Cited by
-
Endoplasmic Reticulum Stress Activates Unfolded Protein Response Signaling and Mediates Inflammation, Obesity, and Cardiac Dysfunction: Therapeutic and Molecular Approach.Front Pharmacol. 2019 Sep 10;10:977. doi: 10.3389/fphar.2019.00977. eCollection 2019. Front Pharmacol. 2019. PMID: 31551782 Free PMC article. Review.
-
Transcriptional Effects of Candidate COVID-19 Treatments on Cardiac Myocytes.Front Cardiovasc Med. 2022 May 24;9:844441. doi: 10.3389/fcvm.2022.844441. eCollection 2022. Front Cardiovasc Med. 2022. PMID: 35686037 Free PMC article.
-
Roles for ATF6 and the sarco/endoplasmic reticulum protein quality control system in the heart.J Mol Cell Cardiol. 2014 Jun;71:11-5. doi: 10.1016/j.yjmcc.2013.09.018. Epub 2013 Oct 16. J Mol Cell Cardiol. 2014. PMID: 24140798 Free PMC article. Review.
-
Paracrine signal emanating from stressed cardiomyocytes aggravates inflammatory microenvironment in diabetic cardiomyopathy.iScience. 2022 Feb 23;25(3):103973. doi: 10.1016/j.isci.2022.103973. eCollection 2022 Mar 18. iScience. 2022. PMID: 35281739 Free PMC article.
-
Hyperglycemia-triggered ATF6-CHOP pathway aggravates acute inflammatory liver injury by β-catenin signaling.Cell Death Discov. 2022 Mar 14;8(1):115. doi: 10.1038/s41420-022-00910-z. Cell Death Discov. 2022. PMID: 35289326 Free PMC article.
References
-
- Porter KR, Kallman FL. Significance of cell particulates as seen by electron microscopy. Ann N Y Acad Sci. 1952;54:882–91. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

