Separate mechanisms act concurrently to shed and release the prion protein from the cell
- PMID: 23093798
- PMCID: PMC3510861
- DOI: 10.4161/pri.22588
Separate mechanisms act concurrently to shed and release the prion protein from the cell
Abstract
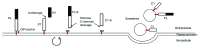
The cellular prion protein (PrP (C) ) is attached to the cell membrane via its glycosylphosphatidylinositol (GPI)-anchor and is constitutively shed into the extracellular space. Here, three different mechanisms are presented that concurrently shed PrP (C) from the cell. The fast α-cleavage released a N-terminal fragment (N1) into the medium and the extreme C-terminal cleavage shed soluble full-length (FL-S) PrP and C-terminally cleaved (C1-S) fragments outside the cell. Also, a slow exosomal release of full-length (FL) and C1-fragment (C1) was demonstrated. The three separate mechanisms acting simultaneously, but with different kinetics, have to be taken into consideration when elucidating functional roles of PrP (C) and also when processing of PrP (C) is considered as a target for intervention in prion diseases. Further, in this study it was shown that metalloprotease inhibitors affected the extreme C-terminal cleavage and shedding of PrP (C) . The metalloprotease inhibitors did not influence the α-cleavage or the exosomal release. Taken together, these results are important for understanding the different mechanisms acting in parallel in the shedding and cleavage of PrP (C) .
Figures
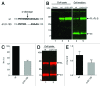
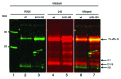

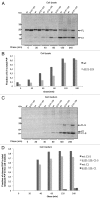
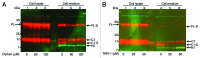
Similar articles
-
Prion protein "gamma-cleavage": characterizing a novel endoproteolytic processing event.Cell Mol Life Sci. 2016 Feb;73(3):667-83. doi: 10.1007/s00018-015-2022-z. Epub 2015 Aug 23. Cell Mol Life Sci. 2016. PMID: 26298290 Free PMC article.
-
Endoproteolytic processing of the mammalian prion glycoprotein family.FEBS J. 2014 Feb;281(3):862-76. doi: 10.1111/febs.12654. Epub 2013 Dec 23. FEBS J. 2014. PMID: 24286250
-
Dual mechanisms for shedding of the cellular prion protein.J Biol Chem. 2004 Mar 19;279(12):11170-8. doi: 10.1074/jbc.M312105200. Epub 2004 Jan 7. J Biol Chem. 2004. PMID: 14711812
-
α-Cleavage of cellular prion protein.Prion. 2012 Nov-Dec;6(5):453-60. doi: 10.4161/pri.22511. Epub 2012 Oct 10. Prion. 2012. PMID: 23052041 Free PMC article. Review.
-
Evolving views in prion glycosylation: functional and pathological implications.Biochimie. 2003 Jan-Feb;85(1-2):33-45. doi: 10.1016/s0300-9084(03)00040-3. Biochimie. 2003. PMID: 12765773 Review.
Cited by
-
Cellular and Molecular Mechanisms of Prion Disease.Annu Rev Pathol. 2019 Jan 24;14:497-516. doi: 10.1146/annurev-pathmechdis-012418-013109. Epub 2018 Oct 24. Annu Rev Pathol. 2019. PMID: 30355150 Free PMC article. Review.
-
Structural and mechanistic aspects influencing the ADAM10-mediated shedding of the prion protein.Mol Neurodegener. 2018 Apr 6;13(1):18. doi: 10.1186/s13024-018-0248-6. Mol Neurodegener. 2018. PMID: 29625583 Free PMC article.
-
Small Extracellular Vesicle-Associated MiRNAs in Polarized Retinal Pigmented Epithelium.Invest Ophthalmol Vis Sci. 2024 Nov 4;65(13):57. doi: 10.1167/iovs.65.13.57. Invest Ophthalmol Vis Sci. 2024. PMID: 39589346 Free PMC article.
-
A seven-residue deletion in PrP leads to generation of a spontaneous prion formed from C-terminal C1 fragment of PrP.J Biol Chem. 2020 Oct 9;295(41):14025-14039. doi: 10.1074/jbc.RA120.014738. Epub 2020 Aug 11. J Biol Chem. 2020. PMID: 32788216 Free PMC article.
-
A new paradigm for enzymatic control of α-cleavage and β-cleavage of the prion protein.J Biol Chem. 2014 Jan 10;289(2):803-13. doi: 10.1074/jbc.M113.502351. Epub 2013 Nov 18. J Biol Chem. 2014. PMID: 24247244 Free PMC article.
References
-
- Harris DA. Cell biological studies of the prion protein. Curr Issues Mol Biol. 1999;1:65–75. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
