Long-term effect of antibodies against infused alpha-galactosidase A in Fabry disease on plasma and urinary (lyso)Gb3 reduction and treatment outcome
- PMID: 23094092
- PMCID: PMC3477102
- DOI: 10.1371/journal.pone.0047805
Long-term effect of antibodies against infused alpha-galactosidase A in Fabry disease on plasma and urinary (lyso)Gb3 reduction and treatment outcome
Abstract
Introduction: Enzyme replacement therapy (ERT) with alpha-Galactosidase A (aGal A) may cause antibody (AB) formation against aGal A in males with Fabry disease (FD). Anti agalsidase ABs negatively influence globotriaosylceramide (Gb3) reduction. We investigated the impact of agalsidase AB on Gb3 and lysoGb3 and clinical outcome in Fabry patients on ERT.
Methods: Adult male and female patients on ERT for at least one year were included. Urinary Gb3 was measured by HPLC, plasma lysoGb3 by LC-ESI-MS/MS and AB with a neutralization assay.
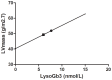
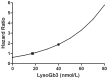
Results: Of the 59 patients evaluable patients, 0/30 females and 17/29 males developed anti-agalsidase antibodies (AB+). Only 3/17 males had transient (low) titers (tolerized). All AB+ patients developed antibodies during the first year of treatment. Change of agalsidase preparation (or dose) did not induce antibody formation. AB+ males had significant less decline in plasma lysoGb3 compared to AB- males (p = 0.04). Urinary Gb3 levels decreased markedly in AB- but remained comparable to baseline in AB+ males (p<0.01). (Lyso)Gb3 reduction in plasma and urine on ERT was correlated with LVmass reduction in females and development white matter lesions and stroke.
Conclusion: In male patients antibodies against aGal A remained present up to 10 years of ERT. The presence of these antibodies is associated with a less robust decrease in plasma lysoGb3 and a profound negative effect on urinary Gb3 reduction, which may reflect worse treatment outcome.
Conflict of interest statement
Figures
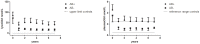

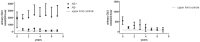
References
-
- Vedder AC, Linthorst GE, van Breemen MJ, Groener JEM, Bemelman FJ, et al. (2007) The Dutch Fabry cohort: diversity of clinical manifestations and Gb3 levels. J Inherit Metab Dis 30(1): 68–78. - PubMed
-
- Eng CM, Guffon N, Wilcox WR, Germain DP, Lee P, et al. (2001) Safety and efficacy of recombinant human alpha-galactosidase A--replacement therapy in Fabry's disease. N Engl J Med 345(1): 9–16. - PubMed
-
- Schiffmann R, Kopp JB, Austin HA, Sabnis S, Moore DF, et al. (2001) Enzyme replacement therapy in Fabry disease: a randomized controlled trial. JAMA 285(21): 2743–2749. - PubMed
-
- Linthorst GE, Hollak CEM, Donker-Koopman WE, Strijland A, Aerts JMFG (2004) Enzyme therapy for Fabry disease: neutralizing antibodies toward agalsidase alpha and beta. Kidney Int 66(4): 1589–1595. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

