Ethanol-mediated facilitation of AMPA receptor function in the dorsomedial striatum: implications for alcohol drinking behavior
- PMID: 23100433
- PMCID: PMC3498079
- DOI: 10.1523/JNEUROSCI.2783-12.2012
Ethanol-mediated facilitation of AMPA receptor function in the dorsomedial striatum: implications for alcohol drinking behavior
Abstract
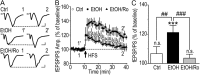
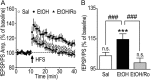
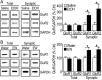
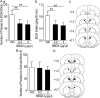
We found previously that acute ex vivo as well as repeated cycles of in vivo ethanol exposure and withdrawal, including excessive voluntary consumption of ethanol, produces a long-lasting increase in the activity of NR2B-containing NMDA receptors (NR2B-NMDARs) in the dorsomedial striatum (DMS) of rats (Wang et al., 2010a). Activation of NMDARs is required for the induction of long-term potentiation (LTP) of AMPA receptor (AMPAR)-mediated synaptic response. We therefore examined whether the ethanol-mediated upregulation of NMDAR activity alters the induction of LTP in the DMS. We found that ex vivo acute exposure of striatal slices to, and withdrawal from, ethanol facilitates the induction of LTP in DMS neurons, which is abolished by the inhibition of NR2B-NMDARs. We also report that repeated systemic administration of ethanol causes an NR2B-NMDAR-dependent facilitation of LTP in the DMS. LTP is mediated by the insertion of AMPAR subunits into the synaptic membrane, and we found that repeated systemic administration of ethanol, as well as cycles of excessive ethanol consumption and withdrawal, produced a long-lasting increase in synaptic localization of the GluR1 and GluR2 subunits of AMPARs in the DMS. Importantly, we report that inhibition of AMPARs in the DMS attenuates operant self-administration of ethanol, but not of sucrose. Together, our data suggest that aberrant synaptic plasticity in the DMS induced by repeated cycles of ethanol exposure and withdrawal contributes to the molecular mechanisms underlying the development and/or maintenance of excessive ethanol consumption.
Figures

References
-
- Barria A, Malinow R. NMDA receptor subunit composition controls synaptic plasticity by regulating binding to CaMKII. Neuron. 2005;48:289–301. - PubMed
-
- Bayer KU, De Koninck P, Leonard AS, Hell JW, Schulman H. Interaction with the NMDA receptor locks CaMKII in an active conformation. Nature. 2001;411:801–805. - PubMed
-
- Belin D, Jonkman S, Dickinson A, Robbins TW, Everitt BJ. Parallel and interactive learning processes within the basal ganglia: relevance for the understanding of addiction. Behav Brain Res. 2009;199:89–102. - PubMed
