A novel internal fixator device for peripheral nerve regeneration
- PMID: 23102114
- PMCID: PMC3629849
- DOI: 10.1089/ten.TEC.2012.0021
A novel internal fixator device for peripheral nerve regeneration
Abstract
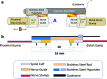
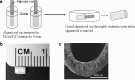
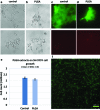
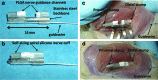
Recovery from peripheral nerve damage, especially for a transected nerve, is rarely complete, resulting in impaired motor function, sensory loss, and chronic pain with inappropriate autonomic responses that seriously impair quality of life. In consequence, strategies for enhancing peripheral nerve repair are of high clinical importance. Tension is a key determinant of neuronal growth and function. In vitro and in vivo experiments have shown that moderate levels of imposed tension (strain) can encourage axonal outgrowth; however, few strategies of peripheral nerve repair emphasize the mechanical environment of the injured nerve. Toward the development of more effective nerve regeneration strategies, we demonstrate the design, fabrication, and implementation of a novel, modular nerve-lengthening device, which allows the imposition of moderate tensile loads in parallel with existing scaffold-based tissue engineering strategies for nerve repair. This concept would enable nerve regeneration in two superposed regimes of nerve extension--traditional extension through axonal outgrowth into a scaffold and extension in intact regions of the proximal nerve, such as that occurring during growth or limb-lengthening. Self-sizing silicone nerve cuffs were fabricated to grip nerve stumps without slippage, and nerves were deformed by actuating a telescoping internal fixator. Poly(lactic co-glycolic) acid (PLGA) constructs mounted on the telescoping rods were apposed to the nerve stumps to guide axonal outgrowth. Neuronal cells were exposed to PLGA using direct contact and extract methods, and they exhibited no signs of cytotoxic effects in terms of cell morphology and viability. We confirmed the feasibility of implanting and actuating our device within a sciatic nerve gap and observed axonal outgrowth following device implantation. The successful fabrication and implementation of our device provides a novel method for examining mechanical influences on nerve regeneration.
Figures


Similar articles
-
Dual-layer conduit containing VEGF-A - Transfected Schwann cells promotes peripheral nerve regeneration via angiogenesis.Acta Biomater. 2024 May;180:323-336. doi: 10.1016/j.actbio.2024.03.029. Epub 2024 Mar 30. Acta Biomater. 2024. PMID: 38561075
-
Engineering a multimodal nerve conduit for repair of injured peripheral nerve.J Neural Eng. 2013 Feb;10(1):016008. doi: 10.1088/1741-2560/10/1/016008. Epub 2013 Jan 3. J Neural Eng. 2013. PMID: 23283383
-
Fabrication of seamless electrospun collagen/PLGA conduits whose walls comprise highly longitudinal aligned nanofibers for nerve regeneration.J Biomed Nanotechnol. 2013 Jun;9(6):931-43. doi: 10.1166/jbn.2013.1605. J Biomed Nanotechnol. 2013. PMID: 23858957
-
Materials for peripheral nerve repair constructs: Natural proteins or synthetic polymers?Neurochem Int. 2021 Feb;143:104953. doi: 10.1016/j.neuint.2020.104953. Epub 2020 Dec 31. Neurochem Int. 2021. PMID: 33388359 Review.
-
The advances in nerve tissue engineering: From fabrication of nerve conduit to in vivo nerve regeneration assays.J Tissue Eng Regen Med. 2019 Nov;13(11):2077-2100. doi: 10.1002/term.2945. Epub 2019 Aug 20. J Tissue Eng Regen Med. 2019. PMID: 31350868 Review.
Cited by
-
Sensory reinnervation of muscle spindles after repair of tibial nerve defects using autogenous vein grafts.Neural Regen Res. 2014 Mar 15;9(6):610-5. doi: 10.4103/1673-5374.130103. Neural Regen Res. 2014. PMID: 25206863 Free PMC article.
-
Peripheral nerve lengthening as a regenerative strategy.Neural Regen Res. 2014 Aug 15;9(16):1498-501. doi: 10.4103/1673-5374.139471. Neural Regen Res. 2014. PMID: 25317163 Free PMC article. Review.
-
Recycle the dental fairy's package: overview of dental pulp stem cells.Stem Cell Res Ther. 2018 Dec 13;9(1):347. doi: 10.1186/s13287-018-1094-8. Stem Cell Res Ther. 2018. PMID: 30545418 Free PMC article. Review.
-
Theory of Bowstring Disease: Diagnosis and Treatment Bowstring Disease.Orthop Surg. 2019 Feb;11(1):3-9. doi: 10.1111/os.12417. Orthop Surg. 2019. PMID: 30834704 Free PMC article. Review.
-
Carriers in cell-based therapies for neurological disorders.Int J Mol Sci. 2014 Jun 13;15(6):10669-723. doi: 10.3390/ijms150610669. Int J Mol Sci. 2014. PMID: 24933636 Free PMC article. Review.
References
-
- Burnett M.G. Zager E.L. Pathophysiology of peripheral nerve injury: a brief review. Neurosurg Focus. 2004;16:E1. - PubMed
-
- Evans G.R. Peripheral nerve injury: a review and approach to tissue engineered constructs. Anat Rec. 2001;263:396. - PubMed
-
- Schmidt C.E. Leach J.B. Neural tissue engineering: strategies for repair and regeneration. Annu Rev Biomed Eng. 2003;5:293. - PubMed
-
- Seddon H.J. Three types of nerve injury. Brain. 1943;66:247.
-
- Sunderland S. A classification of peripheral nerve injury produced by loss of function. Brain. 1951;74:491. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

