The effect of substrate modulus on the growth and function of matrix-embedded endothelial cells
- PMID: 23102623
- PMCID: PMC3505450
- DOI: 10.1016/j.biomaterials.2012.09.079
The effect of substrate modulus on the growth and function of matrix-embedded endothelial cells
Abstract
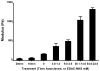
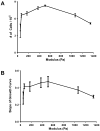
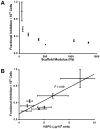
Endothelial cells (EC) are potent bioregulatory cells, modulating thrombosis, inflammation and control over mural smooth muscle cells and vascular health. The biochemical roles of EC are retained when cells are embedded within three-dimensional (3D) denatured collagen matrices. Though substrate mechanics have long been known to affect cellular morphology and function and 3D-EC systems are increasingly used as therapeutic modalities little is known about the effect of substrate mechanics on EC in these 3D systems. In this work, we examined the effect of isolated changes in modulus on EC growth and morphology, extracellular matrix gene expression, modulation of smooth muscle cell growth, and immunogenicity. EC growth, but not morphology was dependent on scaffold modulus. Increased scaffold modulus reduced secretion of smooth muscle cell growth inhibiting heparan sulfate proteoglycans (HSPGs), but had no effect on secreted growth factors, resulting in a loss of smooth muscle cell growth inhibition by EC on high modulus scaffolds. Expression of ICAM-1, VCAM-1 and induction of CD4(+) T cell proliferation was reduced by increased scaffold modulus, and correlated with changes in integrin α5 expression. Expression of several common ECM proteins by EC on stiffer substrates dropped, including collagen IV(α1), collagen IV(α5), fibronectin, HSPGs (perlecan and biglycan). In contrast, expression of elastin and TIMPs were increased. This work shows even modest changes in substrate modulus can have a significant impact on EC function in three-dimensional systems. The mechanism of these changes is not clear, but the data presented here within suggests a model wherein EC attempt to neutralize changes in environmental force balance by altering ECM and integrin expression, leading to changes in effects on downstream signaling and function.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures



References
-
- Yeung T, Georges PC, Flanagan LA, Marg B, Ortiz M, Funaki M, et al. Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion. Cell Motil Cytoskeleton. 2005;60:24–34. - PubMed
-
- Engler AJ, Richert L, Wong JY, Picart C, Discher DE. Surface probe measurements of the elasticity of sectioned tissue, thin gels and polyelectrolyte multilayer films: Correlations between substrate stiffness and cell adhesion. Surface Science. 2004;570:142–54.
-
- Gray DS, Tien J, Chen CS. Repositioning of cells by mechanotaxis on surfaces with micropatterned Young’s modulus. J Biomed Mater Res A. 2003;66:605–14. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

