Retinoic acid induces neurogenesis by activating both retinoic acid receptors (RARs) and peroxisome proliferator-activated receptor β/δ (PPARβ/δ)
- PMID: 23105114
- PMCID: PMC3516764
- DOI: 10.1074/jbc.M112.410381
Retinoic acid induces neurogenesis by activating both retinoic acid receptors (RARs) and peroxisome proliferator-activated receptor β/δ (PPARβ/δ)
Abstract
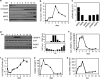
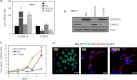
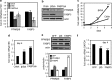
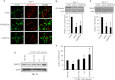
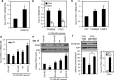
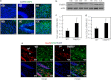
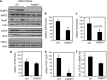
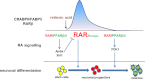
Retinoic acid (RA) regulates gene transcription by activating the nuclear receptors retinoic acid receptor (RAR) and peroxisome proliferator-activated receptor (PPAR) β/δ and their respective cognate lipid-binding proteins CRABP-II and FABP5. RA induces neuronal differentiation, but the contributions of the two transcriptional pathways of the hormone to the process are unknown. Here, we show that the RA-induced commitment of P19 stem cells to neuronal progenitors is mediated by the CRABP-II/RAR path and that the FABP5/PPARβ/δ path can inhibit the process through induction of the RAR repressors SIRT1 and Ajuba. In contrast with its inhibitory activity in the early steps of neurogenesis, the FABP5/PPARβ/δ path promotes differentiation of neuronal progenitors to mature neurons, an activity mediated in part by the PPARβ/δ target gene PDK1. Hence, RA-induced neuronal differentiation is mediated through RAR in the early stages and through PPARβ/δ in the late stages of the process. The switch in RA signaling is accomplished by a transient up-regulation of RARβ concomitantly with a transient increase in the CRABP-II/FABP5 ratio at early stages of differentiation. In accordance with these conclusions, hippocampi of FABP5-null mice display excess accumulation of neuronal progenitor cells and a deficit in mature neurons versus wild-type animals.
Figures

Similar articles
-
All-trans-retinoic acid represses obesity and insulin resistance by activating both peroxisome proliferation-activated receptor beta/delta and retinoic acid receptor.Mol Cell Biol. 2009 Jun;29(12):3286-96. doi: 10.1128/MCB.01742-08. Epub 2009 Apr 13. Mol Cell Biol. 2009. PMID: 19364826 Free PMC article.
-
Overcoming retinoic acid-resistance of mammary carcinomas by diverting retinoic acid from PPARbeta/delta to RAR.Proc Natl Acad Sci U S A. 2008 May 27;105(21):7546-51. doi: 10.1073/pnas.0709981105. Epub 2008 May 21. Proc Natl Acad Sci U S A. 2008. PMID: 18495924 Free PMC article.
-
Stable over-expression of PPARβ/δ and PPARγ to examine receptor signaling in human HaCaT keratinocytes.Cell Signal. 2011 Dec;23(12):2039-50. doi: 10.1016/j.cellsig.2011.07.020. Epub 2011 Aug 4. Cell Signal. 2011. PMID: 21843636 Free PMC article.
-
Non-classical Transcriptional Activity of Retinoic Acid.Subcell Biochem. 2016;81:179-199. doi: 10.1007/978-94-024-0945-1_7. Subcell Biochem. 2016. PMID: 27830505 Review.
-
Retinoic acid as cause of cell proliferation or cell growth inhibition depending on activation of one of two different nuclear receptors.Nutr Rev. 2008 Jan;66(1):55-9. doi: 10.1111/j.1753-4887.2007.00006.x. Nutr Rev. 2008. PMID: 18254885 Review.
Cited by
-
Yokukansan, a Kampo medicine, enhances the level of neuronal lineage markers in differentiated P19 embryonic carcinoma cells.Heliyon. 2019 Oct 16;5(10):e02662. doi: 10.1016/j.heliyon.2019.e02662. eCollection 2019 Oct. Heliyon. 2019. PMID: 31692643 Free PMC article.
-
4-Amino-2-trifluoromethyl-phenyl retinate inhibits proliferation, invasion, and migration of breast cancer cells by independently regulating CRABP2 and FABP5.Drug Des Devel Ther. 2018 Apr 27;12:997-1008. doi: 10.2147/DDDT.S151029. eCollection 2018. Drug Des Devel Ther. 2018. Retraction in: Drug Des Devel Ther. 2024 Jul 03;18:2713-2714. doi: 10.2147/DDDT.S484570. PMID: 29731607 Free PMC article. Retracted.
-
Saturated fatty acids regulate retinoic acid signalling and suppress tumorigenesis by targeting fatty acid-binding protein 5.Nat Commun. 2015 Nov 23;6:8794. doi: 10.1038/ncomms9794. Nat Commun. 2015. PMID: 26592976 Free PMC article.
-
DNA Double-Strand Breaks as Pathogenic Lesions in Neurological Disorders.Int J Mol Sci. 2022 Apr 22;23(9):4653. doi: 10.3390/ijms23094653. Int J Mol Sci. 2022. PMID: 35563044 Free PMC article. Review.
-
Thymus vulgaris Essential Oil in Beta-Cyclodextrin for Solid-State Pharmaceutical Applications.Pharmaceutics. 2023 Mar 11;15(3):914. doi: 10.3390/pharmaceutics15030914. Pharmaceutics. 2023. PMID: 36986775 Free PMC article.
References
-
- Maden M. (2007) Retinoic acid in the development, regeneration, and maintenance of the nervous system. Nat. Rev. Neurosci. 8, 755–765 - PubMed
-
- McBurney M. W., Jones-Villeneuve E. M., Edwards M. K., Anderson P. J. (1982) Control of muscle and neuronal differentiation in a cultured embryonal carcinoma cell line. Nature 299, 165–167 - PubMed
-
- Germain P., Chambon P., Eichele G., Evans R. M., Lazar M. A., Leid M., De Lera A. R., Lotan R., Mangelsdorf D. J., Gronemeyer H. (2006) International Union of Pharmacology. LX. Retinoic acid receptors. Pharmacol. Rev. 58, 712–725 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

