Endocannabinoids via CB₁ receptors act as neurogenic niche cues during cortical development
- PMID: 23108542
- PMCID: PMC3481527
- DOI: 10.1098/rstb.2011.0385
Endocannabinoids via CB₁ receptors act as neurogenic niche cues during cortical development
Abstract
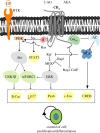
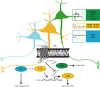
During brain development, neurogenesis is precisely regulated by the concerted action of intrinsic factors and extracellular signalling systems that provide the necessary niche information to proliferating and differentiating cells. A number of recent studies have revealed a previously unknown role for the endocannabinoid (ECB) system in the control of embryonic neuronal development and maturation. Thus, the CB(1) cannabinoid receptor in concert with locally produced ECBs regulates neural progenitor (NP) proliferation, pyramidal specification and axonal navigation. In addition, subcellularly restricted ECB production acts as an axonal growth cone signal to regulate interneuron morphogenesis. These findings provide the rationale for understanding better the consequences of prenatal cannabinoid exposure, and emphasize a novel role of ECBs as neurogenic instructive cues involved in cortical development. In this review the implications of altered CB(1)-receptor-mediated signalling in developmental disorders and particularly in epileptogenesis are briefly discussed.
Figures


References
-
- Sur M., Rubenstein J. L. 2005. Patterning and plasticity of the cerebral cortex. Science 310, 805–81010.1126/science.1112070 (doi:10.1126/science.1112070) - DOI - DOI - PubMed
-
- O'Leary D. D., Chou S. J., Sahara S. 2007. Area patterning of the mammalian cortex. Neuron 56, 252–26910.1016/j.neuron.2007.10.010 (doi:10.1016/j.neuron.2007.10.010) - DOI - DOI - PubMed
-
- Guillemot F., Molnar Z., Tarabykin V., Stoykova A. 2006. Molecular mechanisms of cortical differentiation. Eur. J. Neurosci. 23, 857–86810.1111/j.1460-9568.2006.04626.x (doi:10.1111/j.1460-9568.2006.04626.x) - DOI - DOI - PubMed
-
- Miller F. D., Gauthier A. S. 2007. Timing is everything: making neurons versus glia in the developing cortex. Neuron 54, 357–36910.1016/j.neuron.2007.04.019 (doi:10.1016/j.neuron.2007.04.019) - DOI - DOI - PubMed
-
- Eiraku M., Sasai Y. In press Self-formation of layered neural structures in three-dimensional culture of ES cells. Curr. Opin. Neurobiol. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
