Acid-sensing ion channels (ASICs) in mouse skeletal muscle afferents are heteromers composed of ASIC1a, ASIC2, and ASIC3 subunits
- PMID: 23109675
- PMCID: PMC3545527
- DOI: 10.1096/fj.12-220400
Acid-sensing ion channels (ASICs) in mouse skeletal muscle afferents are heteromers composed of ASIC1a, ASIC2, and ASIC3 subunits
Abstract
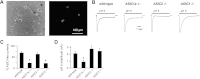
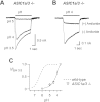
Acid-sensing ion channels (ASICs) are expressed in skeletal muscle afferents, in which they sense extracellular acidosis and other metabolites released during ischemia and exercise. ASICs are formed as homotrimers or heterotrimers of several isoforms (ASIC1a, ASIC1b, ASIC2a, ASIC2b, and ASIC3), with each channel displaying distinct properties. To dissect the ASIC composition in muscle afferents, we used whole-cell patch-clamp recordings to study the properties of acid-evoked currents (amplitude, pH sensitivity, the kinetics of desensitization and recovery from desensitization, and pharmacological modulation) in isolated, labeled mouse muscle afferents from wild-type (C57BL/6J) and specific ASIC(-/-) mice. We found that ASIC-like currents in wild-type muscle afferents displayed fast desensitization, indicating that they are carried by heteromeric channels. Currents from ASIC1a(-/-) muscle afferents were less pH-sensitive and displayed faster recovery, currents from ASIC2(-/-) mice showed diminished potentiation by zinc, and currents from ASIC3(-/-) mice displayed slower desensitization than those from wild-type mice. Finally, ASIC-like currents were absent from triple-null mice lacking ASIC1a, ASIC2a, and ASIC3. We conclude that ASIC1a, ASIC2a, and ASIC3 heteromers are the principle channels in skeletal muscle afferents. These results will help us understand the role of ASICs in exercise physiology and provide a molecular target for potential drug therapies to treat muscle pain.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

