PARP-1 modulation of mTOR signaling in response to a DNA alkylating agent
- PMID: 23110147
- PMCID: PMC3480502
- DOI: 10.1371/journal.pone.0047978
PARP-1 modulation of mTOR signaling in response to a DNA alkylating agent
Abstract
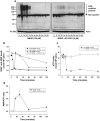
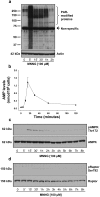
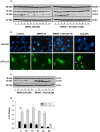
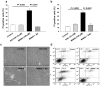
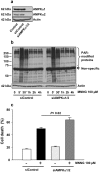
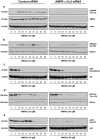
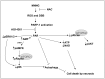
Poly(ADP-ribose) polymerase-1 (PARP-1) is widely involved in cell death responses. Depending on the degree of injury and on cell type, PARP activation may lead to autophagy, apoptosis or necrosis. In HEK293 cells exposed to the alkylating agent N-methyl-N'-nitro-N'-nitrosoguanine (MNNG), we show that PARP-1 activation triggers a necrotic cell death response. The massive poly(ADP-ribose) (PAR) synthesis following PARP-1 activation leads to the modulation of mTORC1 pathway. Shortly after MNNG exposure, NAD⁺ and ATP levels decrease, while AMP levels drastically increase. We characterized at the molecular level the consequences of these altered nucleotide levels. First, AMP-activated protein kinase (AMPK) is activated and the mTORC1 pathway is inhibited by the phosphorylation of Raptor, in an attempt to preserve cellular energy. Phosphorylation of the mTORC1 target S6 is decreased as well as the phosphorylation of the mTORC2 component Rictor on Thr1135. Finally, Akt phosphorylation on Ser473 is lost and then, cell death by necrosis occurs. Inhibition of PARP-1 with the potent PARP inhibitor AG14361 prevents all of these events. Moreover, the antioxidant N-acetyl-L-cysteine (NAC) can also abrogate all the signaling events caused by MNNG exposure suggesting that reactive oxygen species (ROS) production is involved in PARP-1 activation and modulation of mTOR signaling. In this study, we show that PARP-1 activation and PAR synthesis affect the energetic status of cells, inhibit the mTORC1 signaling pathway and possibly modulate the mTORC2 complex affecting cell fate. These results provide new evidence that cell death by necrosis is orchestrated by the balance between several signaling pathways, and that PARP-1 and PAR take part in these events.
Conflict of interest statement
Figures



Similar articles
-
AMPK mediates a pro-survival autophagy downstream of PARP-1 activation in response to DNA alkylating agents.FEBS Lett. 2013 Jan 16;587(2):170-7. doi: 10.1016/j.febslet.2012.11.018. Epub 2012 Nov 29. FEBS Lett. 2013. PMID: 23201261
-
Astrocytic poly(ADP-ribose) polymerase-1 activation leads to bioenergetic depletion and inhibition of glutamate uptake capacity.Glia. 2010 Mar;58(4):446-57. doi: 10.1002/glia.20936. Glia. 2010. PMID: 19795500
-
PARP-1 promotes autophagy via the AMPK/mTOR pathway in CNE-2 human nasopharyngeal carcinoma cells following ionizing radiation, while inhibition of autophagy contributes to the radiation sensitization of CNE-2 cells.Mol Med Rep. 2015 Aug;12(2):1868-76. doi: 10.3892/mmr.2015.3604. Epub 2015 Apr 9. Mol Med Rep. 2015. PMID: 25872765 Free PMC article.
-
Poly(ADP-Ribose)Polymerase 1 (PARP-1) Activation and Ca(2+) Permeable α-Amino-3-Hydroxy-5-Methyl-4-Isoxazolepropionic Acid (AMPA) Channels in Post-Ischemic Brain Damage: New Therapeutic Opportunities?CNS Neurol Disord Drug Targets. 2015;14(5):636-46. doi: 10.2174/1871527314666150430162841. CNS Neurol Disord Drug Targets. 2015. PMID: 25924998 Review.
-
Structure and function of poly(ADP-ribose) polymerase-1: role in oxidative stress-related pathologies.Curr Vasc Pharmacol. 2005 Jul;3(3):209-14. doi: 10.2174/1570161054368625. Curr Vasc Pharmacol. 2005. PMID: 16026317 Review.
Cited by
-
Activation of the PI3K/mTOR Pathway following PARP Inhibition in Small Cell Lung Cancer.PLoS One. 2016 Apr 7;11(4):e0152584. doi: 10.1371/journal.pone.0152584. eCollection 2016. PLoS One. 2016. PMID: 27055253 Free PMC article.
-
Narrative review of emerging roles for AKT-mTOR signaling in cancer radioimmunotherapy.Ann Transl Med. 2021 Oct;9(20):1596. doi: 10.21037/atm-21-4544. Ann Transl Med. 2021. PMID: 34790802 Free PMC article. Review.
-
Glucose deprivation converts poly(ADP-ribose) polymerase-1 hyperactivation into a transient energy-producing process.J Biol Chem. 2013 Dec 20;288(51):36530-7. doi: 10.1074/jbc.M113.506378. Epub 2013 Nov 5. J Biol Chem. 2013. PMID: 24194524 Free PMC article.
-
Potential of Vitamin D and l-Cysteine Co-supplementation to Downregulate Mammalian Target of Rapamycin: A Novel Therapeutic Approach to Diabetes.Metab Syndr Relat Disord. 2025 Feb;23(1):13-22. doi: 10.1089/met.2024.0146. Epub 2024 Sep 16. Metab Syndr Relat Disord. 2025. PMID: 39279596 Review.
-
Intimate Relations-Mitochondria and Ageing.Int J Mol Sci. 2020 Oct 14;21(20):7580. doi: 10.3390/ijms21207580. Int J Mol Sci. 2020. PMID: 33066461 Free PMC article. Review.
References
-
- Ame JC, Spenlehauer C, d Murcia G (2004) The PARP superfamily. Bioessays 26: 882–93. - PubMed
-
- Ethier C, Labelle Y, Poirier GG (2007) PARP-1-induced cell death through inhibition of the MEK/ERK pathway in MNNG-treated HeLa cells. Apoptosis 12: 2037–49. - PubMed
-
- Carling D, Sanders MJ, Woods A (2008) The regulation of AMP-activated protein kinase by upstream kinases. Int J Obes (Lond) 32 Suppl 4S55–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

