A single native ganglioside GM1-binding site is sufficient for cholera toxin to bind to cells and complete the intoxication pathway
- PMID: 23111873
- PMCID: PMC3487775
- DOI: 10.1128/mBio.00401-12
A single native ganglioside GM1-binding site is sufficient for cholera toxin to bind to cells and complete the intoxication pathway
Abstract
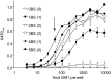
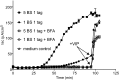
Cholera toxin (CT) from Vibrio cholerae is responsible for the majority of the symptoms of the diarrheal disease cholera. CT is a heterohexameric protein complex with a 240-residue A subunit and a pentameric B subunit of identical 103-residue B polypeptides. The A subunit is proteolytically cleaved within a disulfide-linked loop to generate the A1 and A2 fragments. The B subunit of wild-type (wt) CT binds 5 cell surface ganglioside GM(1) (GM(1)) molecules, and the toxin-GM(1) complex traffics from the plasma membrane (PM) retrograde through endosomes and the Golgi apparatus to the endoplasmic reticulum (ER). From the ER, the enzymatic A1 fragment retrotranslocates to the cytosol to cause disease. Clustering of GM(1) by multivalent toxin binding can structurally remodel cell membranes in ways that may assist toxin uptake and retrograde trafficking. We have recently found, however, that CT may traffic from the PM to the ER by exploiting an endogenous glycosphingolipid pathway (A. A. Wolf et al., Infect. Immun. 76:1476-1484, 2008, and D. J. F. Chinnapen et al., Dev. Cell 23:573-586, 2012), suggesting that multivalent binding to GM(1) is dispensable. Here we formally tested this idea by creating homogenous chimeric holotoxins with defined numbers of native GM(1) binding sites from zero (nonbinding) to five (wild type). We found that a single GM(1) binding site is sufficient for activity of the holotoxin. Therefore, remodeling of cell membranes by mechanisms that involve multivalent binding of toxin to GM(1) receptors is not essential for toxicity of CT. Through multivalent binding to its lipid receptor, cholera toxin (CT) can remodel cell membranes in ways that may assist host cell invasion. We recently found that CT variants which bind no more than 2 receptor molecules do exhibit toxicity, suggesting that CT may be able to enter cells by coopting an endogenous lipid sorting pathway without clustering receptors. We tested this idea directly by using purified variants of CT with zero to five functional receptor-binding sites (BS). One BS enabled CT to intoxicate cells, supporting the conclusion that CT can enter cells by coopting an endogenous lipid-sorting pathway. Although multivalent receptor binding is not essential, it does increase CT toxicity. These findings suggest that achieving higher receptor binding avidity or affecting membrane dynamics by lipid clustering and membrane remodeling may be driving forces for evolution of AB(5) subunit toxins that can bind multivalently to cell membrane lipid receptors.
Figures



References
-
- Ewers H, Helenius A. 2011. Lipid-mediated endocytosis. Cold Spring Harb. Perspect. Biol. 3:a004721 http://dx.doi.org/10.1101/cshperspect.a004721 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 DK084424/DK/NIDDK NIH HHS/United States
- R01 DK053056/DK/NIDDK NIH HHS/United States
- DK48106/DK/NIDDK NIH HHS/United States
- P30 DK034854/DK/NIDDK NIH HHS/United States
- R21 DK090603/DK/NIDDK NIH HHS/United States
- DK34854/DK/NIDDK NIH HHS/United States
- R37 DK048106/DK/NIDDK NIH HHS/United States
- R01 AI031940/AI/NIAID NIH HHS/United States
- R01 AI31940/AI/NIAID NIH HHS/United States
- DK090603/DK/NIDDK NIH HHS/United States
- R01 DK048106/DK/NIDDK NIH HHS/United States
- DK53056/DK/NIDDK NIH HHS/United States
- R56 DK053056/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

