Dietary manipulation and social isolation alter disease progression in a murine model of coronary heart disease
- PMID: 23112879
- PMCID: PMC3480446
- DOI: 10.1371/journal.pone.0047965
Dietary manipulation and social isolation alter disease progression in a murine model of coronary heart disease
Abstract
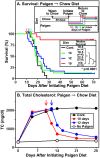
Background: Mice with a deficiency in the HDL receptor SR-BI and low expression of a modified apolipoprotein E gene (SR-BI KO/ApoeR61(h/h)) called 'HypoE' when fed an atherogenic, 'Paigen' diet develop occlusive, atherosclerotic coronary arterial disease (CHD), myocardial infarctions (MI), and heart dysfunction and die prematurely (50% mortality ~40 days after initiation of this diet). Because few murine models share with HypoE mice these cardinal, human-like, features of CHD, HypoE mice represent a novel, small animal, diet-inducible and genetically tractable model for CHD. To better describe the properties of this model, we have explored the effects of varying the composition and timing of administration of atherogenic diets, as well as social isolation vs. group housing, on these animals.
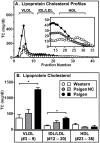
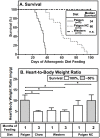
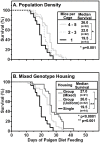
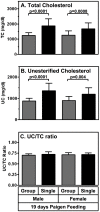
Methodology/principal findings: HypoE mice were maintained on a standard lab chow diet (control) until two months of age. Subsequently they received one of three atherogenic diets (Paigen, Paigen without cholate, Western) or control diet for varying times and were housed in groups or singly, and we determined the plasma cholesterol levels, extent of cardiomegaly and/or survival. The rate of disease progression could be reduced by lowering the severity of the atherogenic diet and accelerated by social isolation. Disease could be induced by Paigen diets either containing or free of cholate. We also established conditions under which CHD could be initiated by an atherogenic diet and then subsequently, by replacing this diet with standard lab chow, hypercholesterolemia could be reduced and progression to early death prevented.
Conclusions/significance: HypoE mice provide a powerful, surgery-free, diet-'titratable' small animal model that can be used to study the onset of recovery from occlusive, atherosclerotic CHD and heart failure due to MI. HypoE mice can be used for the analysis of the effects of environment (diet, social isolation) on a variety of features of cardiovascular disease.
Conflict of interest statement
Figures


Similar articles
-
Diet-induced occlusive coronary atherosclerosis, myocardial infarction, cardiac dysfunction, and premature death in scavenger receptor class B type I-deficient, hypomorphic apolipoprotein ER61 mice.Circulation. 2005 Jun 28;111(25):3457-64. doi: 10.1161/CIRCULATIONAHA.104.523563. Epub 2005 Jun 20. Circulation. 2005. PMID: 15967843
-
Establishment of a novel murine model of ischemic cardiomyopathy with multiple diffuse coronary lesions.PLoS One. 2013 Aug 12;8(8):e70755. doi: 10.1371/journal.pone.0070755. eCollection 2013. PLoS One. 2013. PMID: 23950999 Free PMC article.
-
Loss of PDZK1 causes coronary artery occlusion and myocardial infarction in Paigen diet-fed apolipoprotein E deficient mice.PLoS One. 2009 Dec 1;4(12):e8103. doi: 10.1371/journal.pone.0008103. PLoS One. 2009. PMID: 19956623 Free PMC article.
-
Lowering Targeted Atherogenic Lipoprotein Cholesterol Goals for Patients at "Extreme" ASCVD Risk.Curr Diab Rep. 2019 Nov 21;19(12):146. doi: 10.1007/s11892-019-1246-y. Curr Diab Rep. 2019. PMID: 31754844 Review.
-
Effects of ezetimibe on atherosclerosis in preclinical models.Atherosclerosis. 2011 Apr;215(2):266-78. doi: 10.1016/j.atherosclerosis.2011.02.010. Epub 2011 Feb 17. Atherosclerosis. 2011. PMID: 21397230 Review.
Cited by
-
Identification of apolipoprotein D as a cardioprotective gene using a mouse model of lethal atherosclerotic coronary artery disease.Proc Natl Acad Sci U S A. 2013 Oct 15;110(42):17023-8. doi: 10.1073/pnas.1315986110. Epub 2013 Sep 30. Proc Natl Acad Sci U S A. 2013. PMID: 24082102 Free PMC article.
-
HDL signaling and protection against coronary artery atherosclerosis in mice.J Biomed Res. 2016 Mar;30(2):94-100. doi: 10.7555/JBR.30.20150079. Epub 2015 Aug 20. J Biomed Res. 2016. PMID: 26642235 Free PMC article. Review.
-
Stromal cell-derived factor-1 alpha improves cardiac function in a novel diet-induced coronary atherosclerosis model, the SR-B1ΔCT/LDLR KO mouse.Atherosclerosis. 2024 Aug;395:117518. doi: 10.1016/j.atherosclerosis.2024.117518. Epub 2024 Mar 22. Atherosclerosis. 2024. PMID: 38627162 Free PMC article.
-
One is the deadliest number: the detrimental effects of social isolation on cerebrovascular diseases and cognition.Acta Neuropathol. 2015 Apr;129(4):493-509. doi: 10.1007/s00401-014-1377-9. Epub 2014 Dec 24. Acta Neuropathol. 2015. PMID: 25537401 Free PMC article. Review.
-
Hyperglycemia Aggravates Diet-Induced Coronary Artery Disease and Myocardial Infarction in SR-B1-Knockout/ApoE-Hypomorphic Mice.Front Physiol. 2018 Oct 9;9:1398. doi: 10.3389/fphys.2018.01398. eCollection 2018. Front Physiol. 2018. PMID: 30356742 Free PMC article.
References
-
- Yesilaltay A, Morales MG, Amigo L, Zanlungo S, Rigotti A, et al. (2006) Effects of hepatic expression of the high-density lipoprotein receptor SR-BI on lipoprotein metabolism and female fertility. Endocrinology 147(4): 1577–88. - PubMed
-
- Braun A, Trigatti B, Post M, Sato K, Simons M, et al. (2002) Loss of SR-BI expression leads to the early onset of occlusive atherosclerotic coronary artery disease, spontaneous myocardial infarctions, severe cardiac dysfunction, and premature death in apolipoprotein E-deficient mice. Circ Res 90: 270–276. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

