Myxobacterial tools for social interactions
- PMID: 23123306
- PMCID: PMC3904299
- DOI: 10.1016/j.resmic.2012.10.022
Myxobacterial tools for social interactions
Abstract
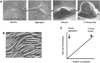
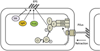
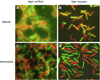
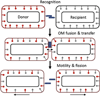
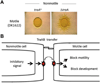
Myxobacteria exhibit complex social traits during which large populations of cells coordinate their behaviors. An iconic example is their response to starvation: thousands of cells move by gliding motility to build a fruiting body in which vegetative cells differentiate into spores. Here we review mechanisms that the model species Myxococcus xanthus uses for cell-cell interactions, with a focus on developmental signaling and social gliding motility. We also discuss a newly discovered cell-cell interaction whereby myxobacteria exchange their outer membrane (OM) proteins and lipids. The mechanism of OM transfer requires physical contact between aligned cells on a hard surface and is apparently mediated by OM fusion. The TraA and TraB proteins are required in both donor and recipient cells for transfer, suggesting bidirectional exchange, and TraA is thought to serve as a cell surface adhesin. OM exchange results in phenotypic changes that can alter gliding motility and development and is proposed to represent a novel microbial interacting platform to coordinate multicellular activities.
Copyright © 2012. Published by Elsevier Masson SAS.
Figures

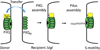

References
-
- Black WP, Xu Q, Yang Z. Type IV pili function upstream of the Dif chemotaxis pathway in Myxococcus xanthus EPS regulation. Mol. Microbiol. 2006;61:447–456. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

