Deficiency in mural vascular cells coincides with blood-brain barrier disruption in Alzheimer's disease
- PMID: 23126372
- PMCID: PMC3628957
- DOI: 10.1111/bpa.12004
Deficiency in mural vascular cells coincides with blood-brain barrier disruption in Alzheimer's disease
Abstract
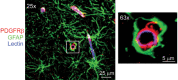
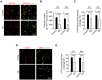
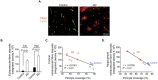
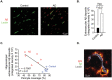
Neurovascular dysfunction contributes to Alzheimer's disease (AD). Cerebrovascular abnormalities and blood-brain barrier (BBB) damage have been shown in AD. The BBB dysfunction can lead to leakage of potentially neurotoxic plasma components in brain that may contribute to neuronal injury. Pericytes are integral in maintaining the BBB integrity. Pericyte-deficient mice develop a chronic BBB damage preceding neuronal injury. Moreover, loss of pericytes was associated with BBB breakdown in patients with amyotrophic lateral sclerosis. Here, we demonstrate a decrease in mural vascular cells in AD, and show that pericyte number and coverage in the cortex and hippocampus of AD subjects compared with neurologically intact controls are reduced by 59% and 60% (P < 0.01), and 32% and 33% (P < 0.01), respectively. An increase in extravascular immunoglobulin G (IgG) and fibrin deposition correlated with reductions in pericyte coverage in AD cases compared with controls; the Pearson's correlation coefficient r for the magnitude of BBB breakdown to IgG and fibrin vs. reduction in pericyte coverage was -0.96 (P < 0.01) and -0.81 (P < 0.01) in the cortex, respectively, and -0.86 (P < 0.01) and -0.98 (P < 0.01) in the hippocampus, respectively. Thus, deficiency in mural vascular cells may contribute to disrupted vascular barrier properties and resultant neuronal dysfunction during AD pathogenesis.
© 2012 The Authors; Brain Pathology © 2012 International Society of Neuropathology.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Armulik A, Genove G, Mae M, Nisancioglu MH, Wallgard E, Niaudet C et al (2010) Pericytes regulate the blood‐brain barrier. Nature 468:557–561. - PubMed
-
- Armulik A, Genove G, Betsholtz C (2011) Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev Cell 21:193–215. - PubMed
-
- Baloyannis SJ, Baloyannis IS (2012) The vascular factor in Alzheimer's disease: a study in Golgi technique and electron microscopy. J Neurol Sci 322:117–121. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

