Mesenchymal stromal cells improve survival during sepsis in the absence of heme oxygenase-1: the importance of neutrophils
- PMID: 23132816
- PMCID: PMC3572335
- DOI: 10.1002/stem.1270
Mesenchymal stromal cells improve survival during sepsis in the absence of heme oxygenase-1: the importance of neutrophils
Abstract
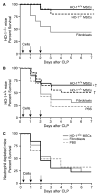
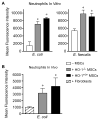
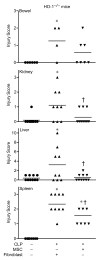
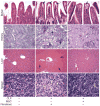
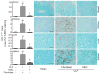
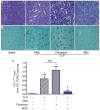
The use of mesenchymal stromal cells (MSCs) for treatment of bacterial infections, including systemic processes like sepsis, is an evolving field of investigation. This study was designed to investigate the potential use of MSCs, harvested from compact bone, and their interactions with the innate immune system, during polymicrobial sepsis induced by cecal ligation and puncture (CLP). We also wanted to elucidate the role of endogenous heme oxygenase (HO)-1 in MSCs during a systemic bacterial infection. MSCs harvested from the bones of HO-1 deficient (-/-) and wild-type (+/+) mice improved the survival of HO-1(-/-) and HO-1(+/+) recipient mice when administered after the onset of polymicrobial sepsis induced by CLP, compared with the administration of fibroblast control cells. The MSCs, originating from compact bone in mice, enhanced the ability of neutrophils to phagocytize bacteria in vitro and in vivo and to promote bacterial clearance in the peritoneum and blood after CLP. Moreover, after depleting neutrophils in recipient mice, the beneficial effects of MSCs were entirely lost, demonstrating the importance of neutrophils for this MSC response. MSCs also decreased multiple organ injury in susceptible HO-1(-/-) mice, when administered after the onset of sepsis. Taken together, these data demonstrate that the beneficial effects of treatment with MSCs after the onset of polymicrobial sepsis is not dependent on endogenous HO-1 expression, and that neutrophils are crucial for this therapeutic response.
Copyright © 2012 AlphaMed Press.
Conflict of interest statement
The authors indicate no potential conflicts of interest.
Figures
References
-
- Friedenstein AJ, Chailakhyan RK, Latsinik NV, et al. Stromal cells responsible for transferring the microenvironment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo. Transplantation. 1974;17:331–340. - PubMed
-
- Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;276:71–74. - PubMed
-
- Dennis JE, Merriam A, Awadallah A, et al. A quadripotential mesenchymal progenitor cell isolated from the marrow of an adult mouse. J Bone Miner Res. 1999;14:700–709. - PubMed
-
- Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. - PubMed
-
- da Silva Meirelles L, Chagastelles PC, Nardi NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci. 2006;119:2204–2213. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

