Neurodevelopmental role for VGLUT2 in pyramidal neuron plasticity, dendritic refinement, and in spatial learning
- PMID: 23136427
- PMCID: PMC3501834
- DOI: 10.1523/JNEUROSCI.4505-11.2012
Neurodevelopmental role for VGLUT2 in pyramidal neuron plasticity, dendritic refinement, and in spatial learning
Abstract
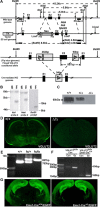
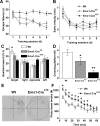
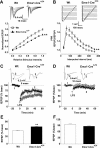
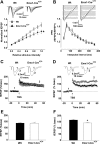
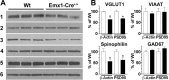
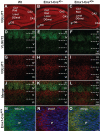
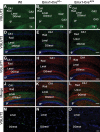
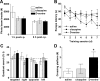
The level and integrity of glutamate transmission during critical periods of postnatal development plays an important role in the refinement of pyramidal neuron dendritic arbor, synaptic plasticity, and cognition. Presently, it is not clear how excitatory transmission via the two predominant isoforms of the vesicular glutamate transporter (VGLUT1 and VGLUT2) participate in this process. To assess a neurodevelopmental role for VGLUT2 in pyramidal neuron maturation, we generated recombinant VGLUT2 knock-out mice and inactivated VGLUT2 throughout development using Emx1-Cre(+/+) knock-in mice. We show that VGLUT2 deficiency in corticolimbic circuits results in reduced evoked glutamate transmission, release probability, and LTD at hippocampal CA3-CA1 synapses during a formative developmental period (postnatal days 11-14). In adults, we find a marked reduction in the amount of dendritic arbor across the span of the dendritic tree of CA1 pyramidal neurons and reduced long-term potentiation and levels of synaptic markers spinophilin and VGLUT1. Loss of dendritic arbor is accompanied by corresponding reductions in the number of dendritic spines, suggesting widespread alterations in synaptic connectivity. Conditional VGLUT2 knock-out mice exhibit increased open-field exploratory activity yet impaired spatial learning and memory, endophenotypes similar to those of NMDA receptor knock-down mice. Remarkably, the impairment in learning can be partially restored by selectively increasing NMDA receptor-mediated glutamate transmission in adult mice by prolonged treatment with d-serine and a d-amino acid oxidase inhibitor. Our data indicate that VGLUT2 expression is pivotal to the proper development of mature pyramidal neuronal architecture and plasticity, and that such glutamatergic deficiency leads to cognitive malfunction as observed in several neurodevelopmental psychiatric disorders.
Figures

Similar articles
-
Homeostatic scaling of vesicular glutamate and GABA transporter expression in rat neocortical circuits.J Neurosci. 2005 Aug 3;25(31):7121-33. doi: 10.1523/JNEUROSCI.5221-04.2005. J Neurosci. 2005. PMID: 16079394 Free PMC article.
-
Vesicular glutamate transporter VGLUT1 has a role in hippocampal long-term potentiation and spatial reversal learning.Cereb Cortex. 2010 Mar;20(3):684-93. doi: 10.1093/cercor/bhp133. Epub 2009 Jul 2. Cereb Cortex. 2010. PMID: 19574394
-
Excitation-transcription coupling via calcium/calmodulin-dependent protein kinase/ERK1/2 signaling mediates the coordinate induction of VGLUT2 and Narp triggered by a prolonged increase in glutamatergic synaptic activity.J Biol Chem. 2010 May 7;285(19):14366-76. doi: 10.1074/jbc.M109.080069. Epub 2010 Mar 8. J Biol Chem. 2010. PMID: 20212045 Free PMC article.
-
Genetic inactivation of the vesicular glutamate transporter 2 (VGLUT2) in the mouse: what have we learnt about functional glutamatergic neurotransmission?Ups J Med Sci. 2010 Feb;115(1):11-20. doi: 10.3109/03009730903572073. Ups J Med Sci. 2010. PMID: 20187846 Free PMC article. Review.
-
Activity-dependent regulation of vesicular glutamate and GABA transporters: a means to scale quantal size.Neurochem Int. 2006 May-Jun;48(6-7):643-9. doi: 10.1016/j.neuint.2005.12.029. Epub 2006 Mar 20. Neurochem Int. 2006. PMID: 16546297 Review.
Cited by
-
BDNF regulates the expression and distribution of vesicular glutamate transporters in cultured hippocampal neurons.PLoS One. 2013;8(1):e53793. doi: 10.1371/journal.pone.0053793. Epub 2013 Jan 11. PLoS One. 2013. PMID: 23326507 Free PMC article.
-
A Mini-Review of the Role of Glutamate Transporter in Drug Addiction.Front Neurol. 2019 Oct 22;10:1123. doi: 10.3389/fneur.2019.01123. eCollection 2019. Front Neurol. 2019. PMID: 31695674 Free PMC article. Review.
-
Differential Contributions of Glutamatergic Hippocampal→Retrosplenial Cortical Projections to the Formation and Persistence of Context Memories.Cereb Cortex. 2019 Jun 1;29(6):2728-2736. doi: 10.1093/cercor/bhy142. Cereb Cortex. 2019. PMID: 29878069 Free PMC article.
-
A Method to Investigate Astrocyte and Microglial Morphological Changes in the Aging Brain of the Rhesus Macaque.Methods Mol Biol. 2019;1938:265-276. doi: 10.1007/978-1-4939-9068-9_19. Methods Mol Biol. 2019. PMID: 30617987 Free PMC article.
-
Research progress on the role of type I vesicular glutamate transporter (VGLUT1) in nervous system diseases.Cell Biosci. 2020 Mar 4;10:26. doi: 10.1186/s13578-020-00393-4. eCollection 2020. Cell Biosci. 2020. PMID: 32158532 Free PMC article. Review.
References
-
- Amitai Y. Thalamocortical synaptic connections: efficacy, modulation, inhibition and plasticity. Rev Neurosci. 2001;12:159–173. - PubMed
-
- Asaka Y, Jugloff DG, Zhang L, Eubanks JH, Fitzsimonds RM. Hippocampal synaptic plasticity is impaired in the Mecp2-null mouse model of Rett syndrome. Neurobiol Dis. 2006;21:217–227. - PubMed
-
- Atkinson L, Batten TF, Moores TS, Varoqui H, Erickson JD, Deuchars J. Differential co-localisation of the P2X7 receptor subunit with vesicular glutamate transporters VGLUT1 and VGLUT2 in rat CNS. Neuroscience. 2004;123:761–768. - PubMed
-
- Balschun D, Moechars D, Callaerts-Vegh Z, Vermaercke B, Van Acker N, Andries L, D'Hooge R. Vesicular glutamate transporter VGLUT1 has a role in hippocampal long-term potentiation and spatial reversal learning. Cereb Cortex. 2010;20:684–693. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
