Micro total analysis systems: fundamental advances and applications in the laboratory, clinic, and field
- PMID: 23140554
- PMCID: PMC3546124
- DOI: 10.1021/ac3031543
Micro total analysis systems: fundamental advances and applications in the laboratory, clinic, and field
Figures

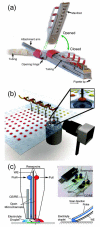





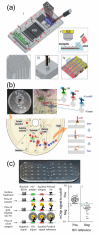


References
-
- Hitzbleck M, Gervais L, Delamarche E. Lab Chip. 2011;11:2673–2679. - PubMed
-
- Vulto P, Podszun S, Meyer P, Hermann C, Manz A, Urban GA. Lab Chip. 2011;11:1596–1602. - PubMed
-
- Luecha J, Hsiao A, Brodsky S, Liu G, Kokini J. Lab Chip. 2011;11:3419–3425. - PubMed
-
- Liu H, Crooks R. J. Am. Chem. Soc. 2011;133:17564–17566. - PubMed
Publication types
MeSH terms
Grants and funding
- F31CA171631/CA/NCI NIH HHS/United States
- R01 EB011763/EB/NIBIB NIH HHS/United States
- F32CA162574/CA/NCI NIH HHS/United States
- F31 CA171631/CA/NCI NIH HHS/United States
- T32 GM007040/GM/NIGMS NIH HHS/United States
- T32GM007040/GM/NIGMS NIH HHS/United States
- K12 GM000678/GM/NIGMS NIH HHS/United States
- K12GM000678/GM/NIGMS NIH HHS/United States
- R01 CA139599/CA/NCI NIH HHS/United States
- F32 CA162574/CA/NCI NIH HHS/United States
- R01 EB012549/EB/NIBIB NIH HHS/United States
- R01EB012549/EB/NIBIB NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

