The frizzled/stan pathway and planar cell polarity in the Drosophila wing
- PMID: 23140623
- PMCID: PMC3575171
- DOI: 10.1016/B978-0-12-394592-1.00001-6
The frizzled/stan pathway and planar cell polarity in the Drosophila wing
Abstract
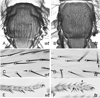
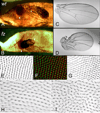
Drosophila has been the key model system for studies on planar cell polarity (PCP). The rich morphology of the insect exoskeleton contains many structures that display PCP. Among these are the trichomes (cuticular hairs) that cover much of the exoskeleton, sensory bristles, and ommatidia. Many genes have been identified that must function for the development of normal PCP. Among these are the genes that comprise the frizzled/starry night (fz/stan) and dachsous/fat pathways. The mechanisms that underlie the function of the fz/stan pathway are best understood. All of the protein products of these genes accumulate asymmetrically in wing cells and there is good evidence that this involves local intercellular signaling between protein complexes on the distal edge of one cell and the juxtaposed proximal edge of its neighbor. It is thought that a feedback system, directed transport, and stabilizing protein-protein interactions mediate the formation of distal and proximal protein complexes. These complexes appear to recruit downstream proteins that function to spatially restrict the activation of the cytoskeleton in wing cells. This leads to the formation of the array of distally pointing hairs found on wings.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures




References
-
- Adler P, Taylor J, Charlton J. The domineering non-autonomy of frizzled and van Gogh clones in the Drosophila wing is a consequence of a disruption in local signaling. Mech Dev. 2000a;96:197–207. - PubMed
-
- Adler PN. Planar signaling and morphogenesis in Drosophila. Dev. Cell. 2002;2:525–535. - PubMed
-
- Adler PN, Charlton J, Liu J. Mutations in the cadherin superfamily member gene dachsous cause a tissue polarity phenotype by altering frizzled signaling. Development. 1998;125:959–968. - PubMed
-
- Adler PN, Krasnow RE, Liu J. Tissue polarity points from cells that have higher Frizzled levels towards cells that have lower Frizzled levels. Curr Biol. 1997;7:940–949. - PubMed
-
- Adler PN, Liu J, Charlton J. Cell size and the morphogenesis of epidermal hairs. Genesis. 2000b;28:82–91. - PubMed
NOTE ADDED IN PROOF
-
- Sagner A, Merkel M, Aigouy B, Gaebel J, Brankatschk M, Julicher F, Eaton S. Establishment of Global Patterns of Planar Polarity during Growth of the Drosophila Wing Epithelium. Current Biology. 2012;22:1296–1301. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

