The deacetylase Sirt6 activates the acetyltransferase GCN5 and suppresses hepatic gluconeogenesis
- PMID: 23142079
- PMCID: PMC3534905
- DOI: 10.1016/j.molcel.2012.09.030
The deacetylase Sirt6 activates the acetyltransferase GCN5 and suppresses hepatic gluconeogenesis
Abstract
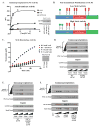
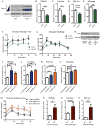
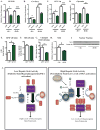
Hepatic glucose production (HGP) maintains blood glucose levels during fasting but can also exacerbate diabetic hyperglycemia. HGP is dynamically controlled by a signaling/transcriptional network that regulates the expression/activity of gluconeogenic enzymes. A key mediator of gluconeogenic gene transcription is PGC-1α. PGC-1α's activation of gluconeogenic gene expression is dependent upon its acetylation state, which is controlled by the acetyltransferase GCN5 and the deacetylase Sirt1. Nevertheless, whether other chromatin modifiers-particularly other sirtuins-can modulate PGC-1α acetylation is currently unknown. Herein, we report that Sirt6 strongly controls PGC-1α acetylation. Surprisingly, Sirt6 induces PGC-1α acetylation and suppresses HGP. Sirt6 depletion decreases PGC-1α acetylation and promotes HGP. These acetylation effects are GCN5 dependent: Sirt6 interacts with and modifies GCN5, enhancing GCN5's activity. Lepr(db/db) mice, an obese/diabetic animal model, exhibit reduced Sirt6 levels; ectopic re-expression suppresses gluconeogenic genes and normalizes glycemia. Activation of hepatic Sirt6 may therefore be therapeutically useful for treating insulin-resistant diabetes.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures




Similar articles
-
CITED2 links hormonal signaling to PGC-1α acetylation in the regulation of gluconeogenesis.Nat Med. 2012 Mar 18;18(4):612-7. doi: 10.1038/nm.2691. Nat Med. 2012. PMID: 22426420
-
The Methionine Transamination Pathway Controls Hepatic Glucose Metabolism through Regulation of the GCN5 Acetyltransferase and the PGC-1α Transcriptional Coactivator.J Biol Chem. 2016 May 13;291(20):10635-45. doi: 10.1074/jbc.M115.706200. Epub 2016 Mar 28. J Biol Chem. 2016. PMID: 27022023 Free PMC article.
-
A fasting inducible switch modulates gluconeogenesis via activator/coactivator exchange.Nature. 2008 Nov 13;456(7219):269-73. doi: 10.1038/nature07349. Epub 2008 Oct 5. Nature. 2008. PMID: 18849969 Free PMC article.
-
GCN5 acetyltransferase in cellular energetic and metabolic processes.Biochim Biophys Acta Gene Regul Mech. 2021 Feb;1864(2):194626. doi: 10.1016/j.bbagrm.2020.194626. Epub 2020 Aug 19. Biochim Biophys Acta Gene Regul Mech. 2021. PMID: 32827753 Free PMC article. Review.
-
Deacetylation of PGC-1α by SIRT1: importance for skeletal muscle function and exercise-induced mitochondrial biogenesis.Appl Physiol Nutr Metab. 2011 Oct;36(5):589-97. doi: 10.1139/h11-070. Epub 2011 Sep 2. Appl Physiol Nutr Metab. 2011. PMID: 21888529 Review.
Cited by
-
O-GlycNacylation Remission Retards the Progression of Non-Alcoholic Fatty Liver Disease.Cells. 2022 Nov 16;11(22):3637. doi: 10.3390/cells11223637. Cells. 2022. PMID: 36429065 Free PMC article. Review.
-
Propargylglycine inhibits hypotaurine/taurine synthesis and elevates cystathionine and homocysteine concentrations in primary mouse hepatocytes.Amino Acids. 2015 Jun;47(6):1215-23. doi: 10.1007/s00726-015-1948-7. Epub 2015 Mar 13. Amino Acids. 2015. PMID: 25772816 Free PMC article.
-
SIRT6 Promotes Hepatic Beta-Oxidation via Activation of PPARα.Cell Rep. 2019 Dec 17;29(12):4127-4143.e8. doi: 10.1016/j.celrep.2019.11.067. Cell Rep. 2019. PMID: 31851938 Free PMC article.
-
Sirt6 reprograms myofibers to oxidative type through CREB-dependent Sox6 suppression.Nat Commun. 2022 Apr 4;13(1):1808. doi: 10.1038/s41467-022-29472-5. Nat Commun. 2022. PMID: 35379817 Free PMC article.
-
Macrophage Polarization and Reprogramming in Acute Inflammation: A Redox Perspective.Antioxidants (Basel). 2022 Jul 19;11(7):1394. doi: 10.3390/antiox11071394. Antioxidants (Basel). 2022. PMID: 35883885 Free PMC article. Review.
References
-
- Coste A, Louet JF, Lagouge M, Lerin C, Antal MC, Meziane H, Schoonjans K, Puigserver P, O’Malley BW, Auwerx J. The genetic ablation of SRC-3 protects against obesity and improves insulin sensitivity by reducing the acetylation of PGC-1{alpha} Proc Natl Acad Sci U S A. 2008;105:17187–17192. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

