Loss-of-function mutations in IGSF1 cause an X-linked syndrome of central hypothyroidism and testicular enlargement
- PMID: 23143598
- PMCID: PMC3511587
- DOI: 10.1038/ng.2453
Loss-of-function mutations in IGSF1 cause an X-linked syndrome of central hypothyroidism and testicular enlargement
Abstract
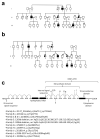
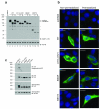
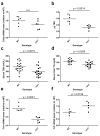
Congenital central hypothyroidism occurs either in isolation or in conjunction with other pituitary hormone deficits. Using exome and candidate gene sequencing, we identified 8 distinct mutations and 2 deletions in IGSF1 in males from 11 unrelated families with central hypothyroidism, testicular enlargement and variably low prolactin concentrations. IGSF1 is a membrane glycoprotein that is highly expressed in the anterior pituitary gland, and the identified mutations impair its trafficking to the cell surface in heterologous cells. Igsf1-deficient male mice show diminished pituitary and serum thyroid-stimulating hormone (TSH) concentrations, reduced pituitary thyrotropin-releasing hormone (TRH) receptor expression, decreased triiodothyronine concentrations and increased body mass. Collectively, our observations delineate a new X-linked disorder in which loss-of-function mutations in IGSF1 cause central hypothyroidism, likely secondary to an associated impairment in pituitary TRH signaling.
Figures

References
-
- Elmlinger MW, Kuhnel W, Lambrecht HG, Ranke MB. Reference intervals from birth to adulthood for serum thyroxine (T4), triiodothyronine (T3), free T3, free T4, thyroxine binding globulin (TBG) and thyrotropin (TSH) Clin Chem. Lab Med. 2001;39:973–979. - PubMed
-
- Mehta A, et al. Is the thyrotropin-releasing hormone test necessary in the diagnosis of central hypothyroidism in children. J Clin Endocrinol Metab. 2003;88:5696–5703. - PubMed
-
- van Tijn DA, de Vijlder JJ, Vulsma T. Role of the thyrotropin-releasing hormone stimulation test in diagnosis of congenital central hypothyroidism in infants. J Clin Endocrinol Metab. 2008;93:410–419. - PubMed
-
- Faglia G, et al. Plasma thyrotropin response to thyrotropin-releasing hormone in patients with pituitary and hypothalamic disorders. J Clin Endocrinol Metab. 1973;37:595–601. - PubMed
-
- Crofton PM, Tepper LA, Kelnar CJ. An evaluation of the thyrotrophin-releasing hormone stimulation test in paediatric clinical practice. Horm Res. 2008;69:53–59. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

