A study of diverse clinical decision support rule authoring environments and requirements for integration
- PMID: 23145874
- PMCID: PMC3554596
- DOI: 10.1186/1472-6947-12-128
A study of diverse clinical decision support rule authoring environments and requirements for integration
Abstract
Background: Efficient rule authoring tools are critical to allow clinical Knowledge Engineers (KEs), Software Engineers (SEs), and Subject Matter Experts (SMEs) to convert medical knowledge into machine executable clinical decision support rules. The goal of this analysis was to identify the critical success factors and challenges of a fully functioning Rule Authoring Environment (RAE) in order to define requirements for a scalable, comprehensive tool to manage enterprise level rules.
Methods: The authors evaluated RAEs in active use across Partners Healthcare, including enterprise wide, ambulatory only, and system specific tools, with a focus on rule editors for reminder and medication rules. We conducted meetings with users of these RAEs to discuss their general experience and perceived advantages and limitations of these tools.
Results: While the overall rule authoring process is similar across the 10 separate RAEs, the system capabilities and architecture vary widely. Most current RAEs limit the ability of the clinical decision support (CDS) interventions to be standardized, sharable, interoperable, and extensible. No existing system meets all requirements defined by knowledge management users.
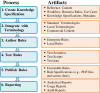
Conclusions: A successful, scalable, integrated rule authoring environment will need to support a number of key requirements and functions in the areas of knowledge representation, metadata, terminology, authoring collaboration, user interface, integration with electronic health record (EHR) systems, testing, and reporting.
Figures


Similar articles
-
Multi-model-based interactive authoring environment for creating shareable medical knowledge.Comput Methods Programs Biomed. 2017 Oct;150:41-72. doi: 10.1016/j.cmpb.2017.07.010. Epub 2017 Jul 22. Comput Methods Programs Biomed. 2017. PMID: 28859829
-
Design and implementation of a standards-based interoperable clinical decision support architecture in the context of the Korean EHR.Int J Med Inform. 2010 Sep;79(9):611-22. doi: 10.1016/j.ijmedinf.2010.06.002. Int J Med Inform. 2010. PMID: 20620098
-
Structured representation for core elements of common clinical decision support interventions to facilitate knowledge sharing.Stud Health Technol Inform. 2013;192:195-9. Stud Health Technol Inform. 2013. PMID: 23920543
-
Review and evaluation of electronic health records-driven phenotype algorithm authoring tools for clinical and translational research.J Am Med Inform Assoc. 2015 Nov;22(6):1251-60. doi: 10.1093/jamia/ocv070. Epub 2015 Jul 29. J Am Med Inform Assoc. 2015. PMID: 26224336 Free PMC article. Review.
-
Decision Support Tools within the Electronic Health Record.Clin Lab Med. 2019 Jun;39(2):197-213. doi: 10.1016/j.cll.2019.01.001. Epub 2019 Mar 28. Clin Lab Med. 2019. PMID: 31036275 Review.
Cited by
-
Retrospective checking of compliance with practice guidelines for acute stroke care: a novel experiment using openEHR's Guideline Definition Language.BMC Med Inform Decis Mak. 2014 May 10;14:39. doi: 10.1186/1472-6947-14-39. BMC Med Inform Decis Mak. 2014. PMID: 24886468 Free PMC article.
-
Reusable Generic Clinical Decision Support System Module for Immunization Recommendations in Resource-Constraint Settings.medRxiv [Preprint]. 2024 Sep 23:2024.09.22.24314152. doi: 10.1101/2024.09.22.24314152. medRxiv. 2024. Update in: AMIA Jt Summits Transl Sci Proc. 2025 Jun 10;2025:395-404. PMID: 39399000 Free PMC article. Updated. Preprint.
-
A Computer-Interpretable Guideline for COVID-19: Rapid Development and Dissemination.JMIR Med Inform. 2020 Oct 1;8(10):e21628. doi: 10.2196/21628. JMIR Med Inform. 2020. PMID: 32931443 Free PMC article.
-
Availability and usage of clinical decision support systems (CDSSs) in office-based primary care settings in the USA.BMJ Health Care Inform. 2019 Dec;26(1):e100015. doi: 10.1136/bmjhci-2019-100015. BMJ Health Care Inform. 2019. PMID: 31818828 Free PMC article.
-
Creating Shareable Clinical Decision Support Rules for a Pharmacogenomics Clinical Guideline Using Structured Knowledge Representation.AMIA Annu Symp Proc. 2015 Nov 5;2015:1985-94. eCollection 2015. AMIA Annu Symp Proc. 2015. PMID: 26958298 Free PMC article.
References
-
- Health Information Technology in the United States. The Information Base for Progress. Robert Wood Johnson Foundation, George Washington University Medical Center, and Institute for Health Policy; 2006.
-
- Part III. Department of Health and Human Services. 45 CFR Part 170: Health Information Technology: initial set of standards, implementation specifications, and certification criteria for electronic health record technology; final rule. Federal register. Services DoHaH. 2010. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

