Gene dosage-dependent association of DYRK1A with the cytoskeleton in the brain and lymphocytes of down syndrome patients
- PMID: 23147510
- PMCID: PMC3511598
- DOI: 10.1097/NEN.0b013e31827733c8
Gene dosage-dependent association of DYRK1A with the cytoskeleton in the brain and lymphocytes of down syndrome patients
Abstract
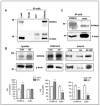
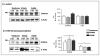
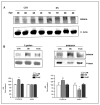
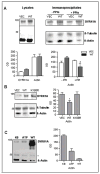
The triplication of the DYRK1A gene encoding proline-directed serine/threonine kinase and located in the critical region of Down syndrome (DS) has been implicated in cognitive deficits and intellectual disability of individuals with DS. We investigated the effect of abnormal levels of this kinase on the cytoskeleton in brain and peripheral tissues of DS subjects. In DS tissues, the predictable approximately equal to 1.5-fold enhancement of the levels of DYRK1A protein was demonstrated. An association of DYRK1A with all 3 major cytoskeleton networks was identified using immunoprecipitation. We concentrated on the actin cytoskeleton because its association with DYRK1A was the most affected by the enzyme levels. As measured by coimmunoprecipitation in DS tissues, but not in fragile X lymphocytes, actin association with DYRK1A was reduced. This reduced association was dependent on the state of phosphorylation of cytoskeletal proteins and was present only in cells overproducing DYRK1A kinase; therefore, the effect was attributable to the DYRK1A gene dosage. Alterations of DYRK1A-actin assemblies were detected in newborn and infant groups, thereby linking DYRK1A overexpression with abnormal brain development of DS children. The identification of the actin cytoskeleton as one of cellular targets of DYRK1A action provides new insights into a gene dosage-sensitive mechanism by which DYRK1A could contribute to the pathogenesis of DS. In addition, the presence of this DS-specific cytoskeleton anomaly in lymphocytes attests to the systemic nature of some features of DS. To our knowledge, this is the first study conducted in human tissue that shows DYRK1A association with the cytoskeleton.
Figures


Similar articles
-
Normalizing the gene dosage of Dyrk1A in a mouse model of Down syndrome rescues several Alzheimer's disease phenotypes.Neurobiol Dis. 2017 Oct;106:76-88. doi: 10.1016/j.nbd.2017.06.010. Epub 2017 Jun 21. Neurobiol Dis. 2017. PMID: 28647555
-
Trisomy-driven overexpression of DYRK1A kinase in the brain of subjects with Down syndrome.Neurosci Lett. 2007 Feb 8;413(1):77-81. doi: 10.1016/j.neulet.2006.11.026. Epub 2006 Dec 4. Neurosci Lett. 2007. PMID: 17145134 Free PMC article.
-
The role of overexpressed DYRK1A protein in the early onset of neurofibrillary degeneration in Down syndrome.Acta Neuropathol. 2008 Oct;116(4):391-407. doi: 10.1007/s00401-008-0419-6. Epub 2008 Aug 12. Acta Neuropathol. 2008. PMID: 18696092 Free PMC article.
-
[Molecular Mechanism Underlying Abnormal Differentiation of Neural Progenitor Cells in the Developing Down Syndrome Brain].Yakugaku Zasshi. 2017;137(7):795-800. doi: 10.1248/yakushi.16-00236-1. Yakugaku Zasshi. 2017. PMID: 28674289 Review. Japanese.
-
DYRK1A: a potential drug target for multiple Down syndrome neuropathologies.CNS Neurol Disord Drug Targets. 2014 Feb;13(1):26-33. doi: 10.2174/18715273113126660186. CNS Neurol Disord Drug Targets. 2014. PMID: 24152332 Review.
Cited by
-
Ten new cases further delineate the syndromic intellectual disability phenotype caused by mutations in DYRK1A.Eur J Hum Genet. 2015 Nov;23(11):1482-7. doi: 10.1038/ejhg.2015.29. Epub 2015 Apr 29. Eur J Hum Genet. 2015. PMID: 25920557 Free PMC article.
-
Role of Microtubule-Associated Protein in Autism Spectrum Disorder.Neurosci Bull. 2018 Dec;34(6):1119-1126. doi: 10.1007/s12264-018-0246-2. Epub 2018 Jun 23. Neurosci Bull. 2018. PMID: 29936584 Free PMC article. Review.
-
Potential Role of Microtubule Stabilizing Agents in Neurodevelopmental Disorders.Int J Mol Sci. 2017 Jul 26;18(8):1627. doi: 10.3390/ijms18081627. Int J Mol Sci. 2017. PMID: 28933765 Free PMC article. Review.
-
New Perspectives of Dyrk1A Role in Neurogenesis and Neuropathologic Features of Down Syndrome.Exp Neurobiol. 2013 Dec;22(4):244-8. doi: 10.5607/en.2013.22.4.244. Epub 2013 Dec 31. Exp Neurobiol. 2013. PMID: 24465139 Free PMC article. Review.
-
DYRK1A in blood and immune function: implications in leukemia, inflammatory disorders, infection and Down syndrome.Front Cell Dev Biol. 2025 May 30;13:1587089. doi: 10.3389/fcell.2025.1587089. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40519271 Free PMC article. Review.
References
-
- Tejedor F, Zhu XR, Kaltenbach E, et al. minibrain: a new protein kinase family involved in postembryonic neurogenesis in Drosophila. Neuron. 1995;14:287–301. - PubMed
-
- Himpel S, Tegge W, Frank R, et al. Specificity determinants of substrate recognition by the protein kinase DYRK1A. J Biol Chem. 2000;275:2431–8. - PubMed
-
- Guimerá J, Casas C, Pucharcòs C, et al. A human homologue of Drosophila minibrain (MNB) is expressed in the neuronal regions affected in Down syndrome and maps to the critical region. Hum Mol Genet. 1996;5:1305–10. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

