Biased agonism of protease-activated receptor 1 by activated protein C caused by noncanonical cleavage at Arg46
- PMID: 23149848
- PMCID: PMC3537315
- DOI: 10.1182/blood-2012-08-452169
Biased agonism of protease-activated receptor 1 by activated protein C caused by noncanonical cleavage at Arg46
Abstract
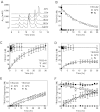
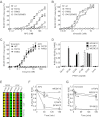
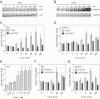
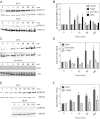
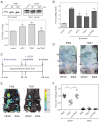
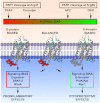
Activated protein C (APC) exerts endothelial cytoprotective actions that require protease-activated receptor 1 (PAR1), whereas thrombin acting via PAR1 causes endothelial disruptive, proinflammatory actions. APC's activities, but not thrombin's, require PAR1 located in caveolae. PAR1 is a biased 7-transmembrane receptor because G proteins mediate thrombin's signaling, whereas β-arrestin 2 mediates APC's signaling. Here we elucidate novel mechanisms for APC's initiation of signaling. Biochemical studies of APC's protease specificity showed that APC cleaved PAR1 sequences at both Arg41 and Arg46. That PAR1 cleavage at Arg46 can occur on cells was supported by APC's cleavage of N-terminal-SEAP-tagged R41Q-PAR1 but not R41Q/R46Q-PAR1 mutants transfected into cells and by anti-PAR1 epitope mapping of APC-treated endothelial cells. A synthetic peptide composing PAR1 residues 47-66, TR47, stimulated protective signaling in endothelial cells as reflected in Akt and glycogen synthase kinase 3β phosphorylation, Ras-related C3 botulinum toxin substrate 1 activation, and barrier stabilization effects. In mice, the TR47 peptide reduced VEGF-induced vascular leakage. These in vitro and in vivo data imply that the novel PAR1 N-terminus beginning at residue Asn47, which is generated by APC cleavage at Arg46, mediates APC's cytoprotective signaling and that this unique APC-generated N-terminal peptide tail is a novel biased agonist for PAR1.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

