p10, the N-terminal domain of p35, protects against CDK5/p25-induced neurotoxicity
- PMID: 23151508
- PMCID: PMC3523839
- DOI: 10.1073/pnas.1212914109
p10, the N-terminal domain of p35, protects against CDK5/p25-induced neurotoxicity
Abstract
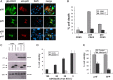
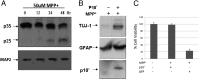
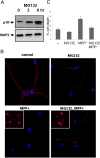
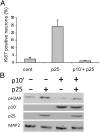
Cyclin-dependent kinase 5(CDK5) in complex with its activator, p35 (protein of 35 kDa), is essential for early neurodevelopment in mammals. However, endogenous cleavage of p35 to p25 is associated with neuron death and neurodegenerative disease. Here we show that a peptide (p10') encoding the N-terminal domain of p35 protects against CDK5/p25-induced toxicity in neurons. p10' also prevented the death of neurons treated with the neurotoxin, 1-methyl-4-phenylpyridinium (MPP(+)), which induces conversion of endogenous p35 to p25, and Parkinson disease (PD)-like symptoms in animals. MPP(+) induces CDK5/p25-dependent phosphorylation of peroxiredoxin 2 (Prx2), resulting in inhibition of its peroxireductase activity and accumulation of reactive oxygen species (ROS). We found that p10' expression inhibited both Prx2 phosphorylation and ROS accumulation in neurons. In addition, p10' inhibited the p25-induced appearance of antigen of the Ki67 antibody (Ki67) and phosphohistone H2AX (γH2AX), classic markers of cell cycle activity and DNA double-strand breakage, respectively, associated with neuron death. Our results suggest that p10 (protein of 10 kDa) is a unique prosurvival domain in p35, essential for normal CDK5/p35 function in neurons. Loss of the p10 domain results in CDK5/p25 toxicity and neurodegeneration in vivo.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
A 24-residue peptide (p5), derived from p35, the Cdk5 neuronal activator, specifically inhibits Cdk5-p25 hyperactivity and tau hyperphosphorylation.J Biol Chem. 2010 Oct 29;285(44):34202-12. doi: 10.1074/jbc.M110.134643. Epub 2010 Aug 18. J Biol Chem. 2010. PMID: 20720012 Free PMC article.
-
The interaction of Munc 18 (p67) with the p10 domain of p35 protects in vivo Cdk5/p35 activity from inhibition by TFP5, a peptide derived from p35.Mol Biol Cell. 2016 Nov 1;27(21):3221-3232. doi: 10.1091/mbc.E15-12-0857. Epub 2016 Sep 14. Mol Biol Cell. 2016. PMID: 27630261 Free PMC article.
-
The protective effects of tanshinone IIA on neurotoxicity induced by β-amyloid protein through calpain and the p35/Cdk5 pathway in primary cortical neurons.Neurochem Int. 2012 Jul;61(2):227-35. doi: 10.1016/j.neuint.2012.04.019. Epub 2012 Apr 25. Neurochem Int. 2012. PMID: 22561406
-
The regulation of cyclin-dependent kinase 5 activity through the metabolism of p35 or p39 Cdk5 activator.Neurosignals. 2003 Sep-Oct;12(4-5):221-9. doi: 10.1159/000074624. Neurosignals. 2003. PMID: 14673209 Review.
-
Peptides derived from Cdk5 activator p35, specifically inhibit deregulated activity of Cdk5.Biotechnol J. 2007 Aug;2(8):978-87. doi: 10.1002/biot.200700057. Biotechnol J. 2007. PMID: 17526058 Review.
Cited by
-
CDK5: A new lead to survival.Cell Cycle. 2013 Jul 1;12(13):1981-2. doi: 10.4161/cc.25304. Epub 2013 Jun 10. Cell Cycle. 2013. PMID: 23759582 Free PMC article. No abstract available.
-
Signals Getting Crossed in the Entanglement of Redox and Phosphorylation Pathways: Phosphorylation of Peroxiredoxin Proteins Sparks Cell Signaling.Antioxidants (Basel). 2019 Jan 23;8(2):29. doi: 10.3390/antiox8020029. Antioxidants (Basel). 2019. PMID: 30678096 Free PMC article. Review.
-
Post-translational modifications of CDK5 and their biological roles in cancer.Mol Biomed. 2021 Jul 20;2(1):22. doi: 10.1186/s43556-021-00029-0. Mol Biomed. 2021. PMID: 35006426 Free PMC article. Review.
-
Focusing on cyclin-dependent kinases 5: A potential target for neurological disorders.Front Mol Neurosci. 2022 Nov 10;15:1030639. doi: 10.3389/fnmol.2022.1030639. eCollection 2022. Front Mol Neurosci. 2022. PMID: 36438186 Free PMC article. Review.
-
Cdk5 Inhibitory Peptide Prevents Loss of Dopaminergic Neurons and Alleviates Behavioral Changes in an MPTP Induced Parkinson's Disease Mouse Model.Front Aging Neurosci. 2018 Jun 1;10:162. doi: 10.3389/fnagi.2018.00162. eCollection 2018. Front Aging Neurosci. 2018. PMID: 29910724 Free PMC article.
References
-
- Ishiguro K, et al. Tau protein kinase I converts normal tau protein into A68-like component of paired helical filaments. J Biol Chem. 1992;267(15):10897–10901. - PubMed
-
- Lew J, Beaudette K, Litwin CM, Wang JH. Purification and characterization of a novel proline-directed protein kinase from bovine brain. J Biol Chem. 1992;267(19):13383–13390. - PubMed
-
- Kobayashi S, et al. A cdc2-related kinase PSSALRE/cdk5 is homologous with the 30 kDa subunit of tau protein kinase II, a proline-directed protein kinase associated with microtubule. FEBS Lett. 1993;335(2):171–175. - PubMed
-
- Lew J, Winkfein RJ, Paudel HK, Wang JH. Brain proline-directed protein kinase is a neurofilament kinase which displays high sequence homology to p34cdc2. J Biol Chem. 1992;267(36):25922–25926. - PubMed
-
- Ishiguro K, et al. Identification of the 23 kDa subunit of tau protein kinase II as a putative activator of cdk5 in bovine brain. FEBS Lett. 1994;342(2):203–208. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

