miR-204 targets Bcl-2 expression and enhances responsiveness of gastric cancer
- PMID: 23152059
- PMCID: PMC3542596
- DOI: 10.1038/cddis.2012.160
miR-204 targets Bcl-2 expression and enhances responsiveness of gastric cancer
Abstract
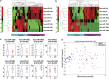
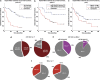
Micro RNAs (miRs) are small non-coding RNAs aberrantly expressed in human tumors. Here, we aim to identify miRs whose deregulated expression leads to the activation of oncogenic pathways in human gastric cancers (GCs). Thirty nine out of 123 tumoral and matched uninvolved peritumoral gastric specimens from three independent European subsets of patients were analyzed for the expression of 851 human miRs using Agilent Platform. The remaining 84 samples were used to validate miRs differentially expressed between tumoral and matched peritumoral specimens by qPCR. miR-204 falls into a group of eight miRs differentially expressed between tumoral and peritumoral samples. Downregulation of miR-204 has prognostic value and correlates with increased staining of Bcl-2 protein in tumoral specimens. Ectopic expression of miR-204 inhibited colony forming ability, migration and tumor engraftment of GC cells. miR-204 targeted Bcl-2 messenger RNA and increased responsiveness of GC cells to 5-fluorouracil and oxaliplatin treatment. Ectopic expression of Bcl-2 protein counteracted miR-204 pro-apoptotic activity in response to 5-fluorouracil. Altogether, these findings suggest that modulation of aberrant expression of miR-204, which in turn releases oncogenic Bcl-2 protein activity might hold promise for preventive and therapeutic strategies of GC.
Figures



Similar articles
-
Her-2 expression regulated by downregulation of miR-9 and which affects chemotherapeutic effect in breast cancer.Cancer Gene Ther. 2017 May;24(5):194-202. doi: 10.1038/cgt.2014.82. Epub 2017 Mar 31. Cancer Gene Ther. 2017. PMID: 28361910
-
Translational control of Bcl-2 promotes apoptosis of gastric carcinoma cells.BMC Cancer. 2021 Jan 5;21(1):12. doi: 10.1186/s12885-020-07711-6. BMC Cancer. 2021. PMID: 33402109 Free PMC article.
-
miR-15b and miR-16 modulate multidrug resistance by targeting BCL2 in human gastric cancer cells.Int J Cancer. 2008 Jul 15;123(2):372-379. doi: 10.1002/ijc.23501. Int J Cancer. 2008. PMID: 18449891
-
miR-135a promotes gastric cancer progression and resistance to oxaliplatin.Oncotarget. 2016 Oct 25;7(43):70699-70714. doi: 10.18632/oncotarget.12208. Oncotarget. 2016. PMID: 27683111 Free PMC article.
-
MicroRNA and cancer--focus on apoptosis.J Cell Mol Med. 2009 Jan;13(1):12-23. doi: 10.1111/j.1582-4934.2008.00510.x. J Cell Mol Med. 2009. PMID: 19175697 Free PMC article. Review.
Cited by
-
LncRNA-UCA1 enhances cell proliferation and 5-fluorouracil resistance in colorectal cancer by inhibiting miR-204-5p.Sci Rep. 2016 Apr 5;6:23892. doi: 10.1038/srep23892. Sci Rep. 2016. PMID: 27046651 Free PMC article.
-
MiRSEA: Discovering the pathways regulated by dysfunctional MicroRNAs.Oncotarget. 2016 Aug 23;7(34):55012-55025. doi: 10.18632/oncotarget.10839. Oncotarget. 2016. PMID: 27474169 Free PMC article.
-
Reprogramming miRNAs global expression orchestrates development of drug resistance in BRAF mutated melanoma.Cell Death Differ. 2019 Jul;26(7):1267-1282. doi: 10.1038/s41418-018-0205-5. Epub 2018 Sep 25. Cell Death Differ. 2019. PMID: 30254376 Free PMC article.
-
The Roles of microRNAs in Cancer Multidrug Resistance.Cancers (Basel). 2022 Feb 21;14(4):1090. doi: 10.3390/cancers14041090. Cancers (Basel). 2022. PMID: 35205839 Free PMC article. Review.
-
Oxibendazole inhibits prostate cancer cell growth.Oncol Lett. 2018 Feb;15(2):2218-2226. doi: 10.3892/ol.2017.7579. Epub 2017 Dec 11. Oncol Lett. 2018. PMID: 29434928 Free PMC article.
References
-
- Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. - PubMed
-
- Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. - PubMed
-
- Calin GA, Croce CM. MicroRNA signatures in human cancers. Nat Rev Cancer. 2006;6:857–866. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

