LRP1 in brain vascular smooth muscle cells mediates local clearance of Alzheimer's amyloid-β
- PMID: 23152628
- PMCID: PMC3508699
- DOI: 10.1523/JNEUROSCI.3987-12.2012
LRP1 in brain vascular smooth muscle cells mediates local clearance of Alzheimer's amyloid-β
Abstract
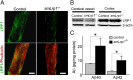
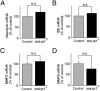
Impaired clearance of amyloid-β (Aβ) is a major pathogenic event for Alzheimer's disease (AD). Aβ depositions in brain parenchyma as senile plaques and along cerebrovasculature as cerebral amyloid angiopathy (CAA) are hallmarks of AD. A major pathway that mediates brain Aβ clearance is the cerebrovascular system where Aβ is eliminated through the blood-brain barrier (BBB) and/or degraded by cerebrovascular cells along the interstitial fluid drainage pathway. An Aβ clearance receptor, the low-density lipoprotein receptor-related protein 1 (LRP1), is abundantly expressed in cerebrovasculature, in particular in vascular smooth muscle cells. Previous studies have indicated a role of LRP1 in endothelial cells in transcytosing Aβ out of the brain across the BBB; however, whether this represents a significant pathway for brain Aβ clearance remains controversial. Here, we demonstrate that Aβ can be cleared locally in the cerebrovasculature by an LRP1-dependent endocytic pathway in smooth muscle cells. The uptake and degradation of both endogenous and exogenous Aβ were significantly reduced in LRP1-suppressed human brain vascular smooth muscle cells. Conditional deletion of Lrp1 in vascular smooth muscle cell in amyloid model APP/PS1 mice accelerated brain Aβ accumulation and exacerbated Aβ deposition as amyloid plaques and CAA without affecting Aβ production. Our results demonstrate that LRP1 is a major Aβ clearance receptor in cerebral vascular smooth muscle cell and a disturbance of this pathway contributes to Aβ accumulation. These studies establish critical functions of the cerebrovasculature system in Aβ metabolism and identify a new pathway involved in the pathogenesis of both AD and CAA.
Figures






References
-
- Borchelt DR, Ratovitski T, van Lare J, Lee MK, Gonzales V, Jenkins NA, Copeland NG, Price DL, Sisodia SS. Accelerated amyloid deposition in the brains of transgenic mice coexpressing mutant presenilin 1 and amyloid precursor proteins. Neuron. 1997;19:939–945. - PubMed
-
- Bu G, Maksymovitch EA, Nerbonne JM, Schwartz AL. Expression and function of the low density lipoprotein receptor-related protein (LRP) in mammalian central neurons. J Biol Chem. 1994;269:18521–18528. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
