How should debriefing be undertaken in web-based studies? Findings from a randomized controlled trial
- PMID: 23160103
- PMCID: PMC3510731
- DOI: 10.2196/jmir.2186
How should debriefing be undertaken in web-based studies? Findings from a randomized controlled trial
Abstract
Background: Internet research may raise older ethical issues in new forms or pose new issues. It has been recommended that debriefing information online be kept very short, with further information including study results made available if requested by participants. There are no empirical studies that compare possible alternative methods of debriefing in online studies.
Objective: To undertake a randomized controlled trial evaluating how to implement the recommended approach by assessing the effects of two different approaches on accessing of additional information.
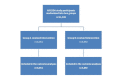
Methods: All 11,943 participants in the Effects of Study Design and Allocation (ESDA) study, which employed deception, were randomly assigned to one of two methods of debriefing: Group A received the debriefing information in the body of an email with links to protocol and results pages; Group B was presented with these links after clicking on an initial link in the body of the email to view the debriefing information on a website. Outcomes assessed were the proportions clicking on the links to the protocol and results summary and the time spent on these pages by those accessing them.
Results: The group who were presented with no debriefing information in the body of the email and went to a website for this information (Group B) were approximately twice as likely to subsequently access the protocol and the results summary. These differences between the two groups were highly statistically significant. Although these differences are clear, the overall proportions accessing such information were low, and there were no differences in mean time spent reading these pages. Only one quarter of Group B actually accessed debriefing information.
Conclusions: In circumstances where the uptake of fuller information on study design, methods, and findings is deemed important, debriefing information may be better provided via a link and not included in the body of an email. Doing so may, however, reduce the extent of receiving any debriefing information at all. There is a wider need for high quality empirical studies to inform ethical evaluations.
Trial registration: Australian New Zealand Clinical Trials Registry, ACTRN12610000846022 (http://www.anzctr.org.au/).
Conflict of interest statement
None declared.
Similar articles
-
Which online format is most effective for assisting Baby Boomers to complete advance directives? A randomised controlled trial of email prompting versus online education module.BMC Palliat Care. 2017 Aug 29;16(1):43. doi: 10.1186/s12904-017-0225-9. BMC Palliat Care. 2017. PMID: 28851337 Free PMC article. Clinical Trial.
-
Effectiveness of a Web 2.0 Intervention to Increase Physical Activity in Real-World Settings: Randomized Ecological Trial.J Med Internet Res. 2017 Nov 13;19(11):e390. doi: 10.2196/jmir.8484. J Med Internet Res. 2017. PMID: 29133282 Free PMC article. Clinical Trial.
-
Demographic, behavioral, and psychosocial correlates of using the website component of a worksite physical activity and healthy nutrition promotion program: a longitudinal study.J Med Internet Res. 2010 Sep 30;12(3):e44. doi: 10.2196/jmir.1402. J Med Internet Res. 2010. PMID: 20921001 Free PMC article.
-
The influence of user characteristics and a periodic email prompt on exposure to an internet-delivered computer-tailored lifestyle program.J Med Internet Res. 2012 Mar 1;14(2):e40. doi: 10.2196/jmir.1939. J Med Internet Res. 2012. PMID: 22382037 Free PMC article. Clinical Trial.
-
A Web-Based, Social Networking Beginners' Running Intervention for Adults Aged 18 to 50 Years Delivered via a Facebook Group: Randomized Controlled Trial.J Med Internet Res. 2018 Feb 26;20(2):e67. doi: 10.2196/jmir.7862. J Med Internet Res. 2018. PMID: 29483065 Free PMC article. Clinical Trial.
Cited by
-
Eliciting debriefing experiences: A scoping review.Heliyon. 2024 Jun 25;10(13):e33592. doi: 10.1016/j.heliyon.2024.e33592. eCollection 2024 Jul 15. Heliyon. 2024. PMID: 39035517 Free PMC article.
-
In randomization we trust? There are overlooked problems in experimenting with people in behavioral intervention trials.J Clin Epidemiol. 2014 Mar;67(3):247-53. doi: 10.1016/j.jclinepi.2013.09.004. Epub 2013 Dec 4. J Clin Epidemiol. 2014. PMID: 24314401 Free PMC article.
-
Effects of study design and allocation on self-reported alcohol consumption: randomized trial.Trials. 2015 Mar 28;16:127. doi: 10.1186/s13063-015-0642-0. Trials. 2015. PMID: 25872651 Free PMC article. Clinical Trial.
-
The use of deception in public health behavioral intervention trials: a case study of three online alcohol trials.Am J Bioeth. 2013;13(11):39-47. doi: 10.1080/15265161.2013.839751. Am J Bioeth. 2013. PMID: 24161181 Free PMC article.
-
Randomized study of two different consent procedures on recall: a study within a digital alcohol intervention trial.Trials. 2024 Jan 2;25(1):5. doi: 10.1186/s13063-023-07855-3. Trials. 2024. PMID: 38167493 Free PMC article. Clinical Trial.
References
-
- Sharkey S, Jones R, Smithson J, Hewis E, Emmens T, Ford T, Owens C. Ethical practice in internet research involving vulnerable people: lessons from a self-harm discussion forum study (SharpTalk) J Med Ethics. 2011 Dec;37(12):752–8. doi: 10.1136/medethics-2011-100080.medethics-2011-100080 - DOI - PubMed
-
- Thompson LA, Black E, Duff WP, Paradise Black N, Saliba H, Dawson K. Protected health information on social networking sites: ethical and legal considerations. J Med Internet Res. 2011;13(1):e8. doi: 10.2196/jmir.1590. http://www.jmir.org/2011/1/e8/v13i1e8 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources