Unlike bone, cartilage regeneration remains elusive
- PMID: 23161992
- PMCID: PMC4327988
- DOI: 10.1126/science.1222454
Unlike bone, cartilage regeneration remains elusive
Abstract
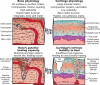
Articular cartilage was predicted to be one of the first tissues to successfully be regenerated, but this proved incorrect. In contrast, bone (but also vasculature and cardiac tissues) has seen numerous successful reparative approaches, despite consisting of multiple cell and tissue types and, thus, possessing more complex design requirements. Here, we use bone-regeneration successes to highlight cartilage-regeneration challenges: such as selecting appropriate cell sources and scaffolds, creating biomechanically suitable tissues, and integrating to native tissue. We also discuss technologies that can address the hurdles of engineering a tissue possessing mechanical properties that are unmatched in human-made materials and functioning in environments unfavorable to neotissue growth.
Figures


References
-
- Langer R, Vacanti JP. Science. 1993 May 14;260:920. - PubMed
-
- Viola J, Lal B, Grad O. National Science Foundation; Arlington, VA: Oct, 2003.
-
- Athanasiou KA, Darling EM, Hu JC. Synthesis Lectures on Tissue Engineering. 2009;1:1. 2009/01/01.
-
- Meyer U, Wiesmann HP. Bone and cartilage engineering. Springer; Berlin ; New York: 2006. pp. xiii–264.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

