Plasma Norepinephrine in Hypertensive Rats Reflects α(2)-Adrenoceptor Release Control Only When Re-Uptake is Inhibited
- PMID: 23162530
- PMCID: PMC3492874
- DOI: 10.3389/fneur.2012.00160
Plasma Norepinephrine in Hypertensive Rats Reflects α(2)-Adrenoceptor Release Control Only When Re-Uptake is Inhibited
Abstract
α(2)-adrenoceptors (AR) lower central sympathetic output and peripheral catecholamine release, thereby protecting against sympathetic hyperactivity and hypertension. Norepinephrine re-uptake-transporter effectively (NET) removes norepinephrine from the synapse. Overflow to plasma will therefore not reflect release. Here we tested if inhibition of re-uptake allowed presynaptic α(2)AR release control to be reflected as differences in norepinephrine overflow in anesthetized hypertensive spontaneously hypertensive rats (SHR) and normotensive rats (WKY). We also tested if α(2)AR modulated the experiment-induced epinephrine secretion, and a phenylephrine-induced, α(1)-adrenergic vasoconstriction. Blood pressure was recorded through a femoral artery catheter, and cardiac output by ascending aorta flow. After pre-treatment with NET inhibitor (desipramine), and/or α(2)AR antagonist (yohimbine, L-659,066) or agonist (clonidine, ST-91), we injected phenylephrine. Arterial blood was sampled 15 min later. Plasma catecholamine concentrations were not influenced by phenylephrine, and therefore reflected effects of pre-treatment. Desipramine and α(2)AR antagonist separately had little effect on norepinephrine overflow. Combined, they increased norepinephrine overflow, particularly in SHR. Clonidine, but not ST-91, reduced, and pertussis toxin increased norepinephrine overflow in SHR and epinephrine secretion in both strains. L-659,066 + clonidine (central α(2)AR-stimulation) normalized the high blood pressure, heart rate, and vascular tension in SHR. α(2)AR antagonists reduced phenylephrine-induced vasoconstriction equally in WKY and SHR.
Conclusions: α(2A)AR inhibition increased norepinephrine overflow only when re-uptake was blocked, and then with particular efficacy in SHR, possibly due to their high sympathetic tone. α(2A)AR inhibited epinephrine secretion, particularly in SHR. α(2A)AR supported α(1)AR-induced vasoconstriction equally in the two strains. α(2)AR malfunctions were therefore not detected in SHR under this basal condition.
Keywords: catecholamine release; epinephrine; hypertension; norepinephrine; norepinephrine re-uptake transporter; plasma catecholamine concentrations; sympathetic nervous system activity; α2-adrenoceptors.
Figures
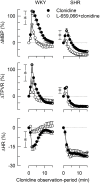
 within symbols) and group differences at peak response (*in brackets left of curves) and at 15 min (*in brackets right of curves) were detected as indicated. Cardiovascular baselines prior to clonidine are shown in Table 2.
within symbols) and group differences at peak response (*in brackets left of curves) and at 15 min (*in brackets right of curves) were detected as indicated. Cardiovascular baselines prior to clonidine are shown in Table 2.  ,*P ≤ 0.025.
,*P ≤ 0.025.
 Within columns; significant responses. *In brackets; significant group differences.
Within columns; significant responses. *In brackets; significant group differences.  P ≤ 0.005; *P ≤ 0.0042.
P ≤ 0.005; *P ≤ 0.0042.Similar articles
-
β- and α2-Adrenoceptor Control of Vascular Tension and Catecholamine Release in Female Normotensive and Spontaneously Hypertensive Rats.Front Neurol. 2017 Apr 3;8:130. doi: 10.3389/fneur.2017.00130. eCollection 2017. Front Neurol. 2017. PMID: 28424658 Free PMC article.
-
α2-Adrenoreceptor Constraint of Catecholamine Release and Blood Pressure Is Enhanced in Female Spontaneously Hypertensive Rats.Front Neurosci. 2016 Mar 30;10:130. doi: 10.3389/fnins.2016.00130. eCollection 2016. Front Neurosci. 2016. PMID: 27065790 Free PMC article.
-
Angiotensin AT1 - α2C-Adrenoceptor Interaction Disturbs α2A-auto-Inhibition of Catecholamine Release in Hypertensive Rats.Front Neurol. 2013 Jun 10;4:70. doi: 10.3389/fneur.2013.00070. eCollection 2013. Front Neurol. 2013. PMID: 23772221 Free PMC article.
-
Altered β1-3-adrenoceptor influence on α2-adrenoceptor-mediated control of catecholamine release and vascular tension in hypertensive rats.Front Physiol. 2015 Apr 20;6:120. doi: 10.3389/fphys.2015.00120. eCollection 2015. Front Physiol. 2015. PMID: 25941491 Free PMC article.
-
Tyramine Reveals Failing α2-Adrenoceptor Control of Catecholamine Release and Total Peripheral Vascular Resistance in Hypertensive Rats.Front Neurol. 2013 Feb 28;4:19. doi: 10.3389/fneur.2013.00019. eCollection 2013. Front Neurol. 2013. PMID: 23450822 Free PMC article.
Cited by
-
Ethanol and liver: recent insights into the mechanisms of ethanol-induced fatty liver.World J Gastroenterol. 2014 Oct 28;20(40):14672-85. doi: 10.3748/wjg.v20.i40.14672. World J Gastroenterol. 2014. PMID: 25356030 Free PMC article. Review.
-
Loss of Group II Metabotropic Glutamate Receptor Signaling Exacerbates Hypertension in Spontaneously Hypertensive Rats.Life (Basel). 2021 Jul 20;11(7):720. doi: 10.3390/life11070720. Life (Basel). 2021. PMID: 34357092 Free PMC article.
-
β- and α2-Adrenoceptor Control of Vascular Tension and Catecholamine Release in Female Normotensive and Spontaneously Hypertensive Rats.Front Neurol. 2017 Apr 3;8:130. doi: 10.3389/fneur.2017.00130. eCollection 2017. Front Neurol. 2017. PMID: 28424658 Free PMC article.
-
β1-Blockers Lower Norepinephrine Release by Inhibiting Presynaptic, Facilitating β1-Adrenoceptors in Normotensive and Hypertensive Rats.Front Neurol. 2014 Apr 16;5:51. doi: 10.3389/fneur.2014.00051. eCollection 2014. Front Neurol. 2014. PMID: 24795691 Free PMC article.
-
α2-Adrenoreceptor Constraint of Catecholamine Release and Blood Pressure Is Enhanced in Female Spontaneously Hypertensive Rats.Front Neurosci. 2016 Mar 30;10:130. doi: 10.3389/fnins.2016.00130. eCollection 2016. Front Neurosci. 2016. PMID: 27065790 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials

