Novel roles of PAK1 in the heart
- PMID: 23162741
- PMCID: PMC3490967
- DOI: 10.4161/cl.21497
Novel roles of PAK1 in the heart
Abstract
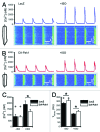
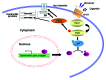
Our work and others' over the past few years have led to the identification of new roles of PAK1 in cardiac physiology, such as the regulation of cardiac ion channel and actomyosin function. More recent studies have revealed that PAK1-deficient mice were vulnerable to cardiac hypertrophy and readily progress to failure under sustained pressure overload and susceptible to ischemia/reperfusion injury. Our further study indicated that the PAK1 activator FTY720 was able to prevent this pressure overload-induced hypertrophy in wild-type mice without compromising their cardiac functions. A cardiac protective effect against ischemia/reperfusion injury by FTY720 was also observed in both rat and mouse models by us and others. Thus, these studies suggest that PAK1 is more important in the heart than previously thought, in particular a therapeutic potential of PAK1 activators. In the future, in-depth investigations are required to further substantiate our hypotheses on mechanisms for PAK1 function in the heart and to explore a therapeutic potential of FTY720 and other PAK1 activators in heart disease conditions.
Figures



Similar articles
-
Pak1 as a novel therapeutic target for antihypertrophic treatment in the heart.Circulation. 2011 Dec 13;124(24):2702-15. doi: 10.1161/CIRCULATIONAHA.111.048785. Epub 2011 Nov 14. Circulation. 2011. PMID: 22082674 Free PMC article.
-
FTY720 prevents ischemia/reperfusion injury-associated arrhythmias in an ex vivo rat heart model via activation of Pak1/Akt signaling.J Mol Cell Cardiol. 2010 Feb;48(2):406-14. doi: 10.1016/j.yjmcc.2009.10.009. Epub 2009 Oct 21. J Mol Cell Cardiol. 2010. PMID: 19852968 Free PMC article.
-
Activation of Pak1/Akt/eNOS signaling following sphingosine-1-phosphate release as part of a mechanism protecting cardiomyocytes against ischemic cell injury.Am J Physiol Heart Circ Physiol. 2011 Oct;301(4):H1487-95. doi: 10.1152/ajpheart.01003.2010. Epub 2011 Jun 24. Am J Physiol Heart Circ Physiol. 2011. PMID: 21705677 Free PMC article.
-
The p21-activated kinase 1 (Pak1) signalling pathway in cardiac disease: from mechanistic study to therapeutic exploration.Br J Pharmacol. 2018 Apr;175(8):1362-1374. doi: 10.1111/bph.13872. Epub 2017 Jun 28. Br J Pharmacol. 2018. PMID: 28574147 Free PMC article. Review.
-
Novel insights into mechanisms for Pak1-mediated regulation of cardiac Ca(2+) homeostasis.Front Physiol. 2015 Mar 17;6:76. doi: 10.3389/fphys.2015.00076. eCollection 2015. Front Physiol. 2015. PMID: 25852566 Free PMC article. Review.
Cited by
-
The PAKs come of age: Celebrating 18 years of discovery.Cell Logist. 2012 Apr 1;2(2):54-58. doi: 10.4161/cl.22084. Cell Logist. 2012. PMID: 23125949 Free PMC article.
-
Murine Electrophysiological Models of Cardiac Arrhythmogenesis.Physiol Rev. 2017 Jan;97(1):283-409. doi: 10.1152/physrev.00007.2016. Physiol Rev. 2017. PMID: 27974512 Free PMC article. Review.
-
PAK1 and Therapy Resistance in Melanoma.Cells. 2023 Sep 28;12(19):2373. doi: 10.3390/cells12192373. Cells. 2023. PMID: 37830586 Free PMC article. Review.
-
Role of p21-activated kinases in cardiovascular development and function.Cell Mol Life Sci. 2013 Nov;70(22):4223-8. doi: 10.1007/s00018-013-1347-8. Epub 2013 May 4. Cell Mol Life Sci. 2013. PMID: 23640572 Free PMC article. Review.
-
MicroRNA-145 targets in cancer and the cardiovascular system: evidence for common signaling pathways.Vasc Biol. 2020 Oct 23;2(1):R115-R128. doi: 10.1530/VB-20-0012. eCollection 2020. Vasc Biol. 2020. PMID: 33283158 Free PMC article. Review.
References
-
- Ke Y, Lei M, Collins TP, Rakovic S, Mattick PA, Yamasaki M, et al. Regulation of L-type calcium channel and delayed rectifier potassium channel activity by p21-activated kinase-1 in guinea pig sinoatrial node pacemaker cells. Circ Res. 2007;100:1317–27. doi: 10.1161/01.RES.0000266742.51389.a4. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
