CLC-3 chloride channels moderate long-term potentiation at Schaffer collateral-CA1 synapses
- PMID: 23165767
- PMCID: PMC3591711
- DOI: 10.1113/jphysiol.2012.243485
CLC-3 chloride channels moderate long-term potentiation at Schaffer collateral-CA1 synapses
Abstract
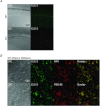
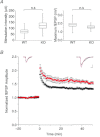
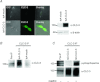
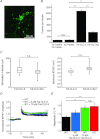
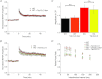
The chloride channel CLC-3 is expressed in the brain on synaptic vesicles and postsynaptic membranes. Although CLC-3 is broadly expressed throughout the brain, the CLC-3 knockout mouse shows complete, selective postnatal neurodegeneration of the hippocampus, suggesting a crucial role for the channel in maintaining normal brain function. CLC-3 channels are functionally linked to NMDA receptors in the hippocampus; NMDA receptor-dependent Ca(2+) entry, activation of Ca(2+)/calmodulin kinase II and subsequent gating of CLC-3 link the channels via a Ca(2+)-mediated feedback loop. We demonstrate that loss of CLC-3 at mature synapses increases long-term potentiation from 135 ± 4% in the wild-type slice preparation to 154 ± 7% above baseline (P < 0.001) in the knockout; therefore, the contribution of CLC-3 is to reduce synaptic potentiation by ∼40%. Using a decoy peptide representing the Ca(2+)/calmodulin kinase II phosphorylation site on CLC-3, we show that phosphorylation of CLC-3 is required for its regulatory function in long-term potentiation. CLC-3 is also expressed on synaptic vesicles; however, our data suggest functionally separable pre- and postsynaptic roles. Thus, CLC-3 confers Cl(-) sensitivity to excitatory synapses, controls the magnitude of long-term potentiation and may provide a protective limit on Ca(2+) influx.
Figures

Similar articles
-
Activation of Group II Metabotropic Glutamate Receptors Promotes LTP Induction at Schaffer Collateral-CA1 Pyramidal Cell Synapses by Priming NMDA Receptors.J Neurosci. 2016 Nov 9;36(45):11521-11531. doi: 10.1523/JNEUROSCI.1519-16.2016. J Neurosci. 2016. PMID: 27911756 Free PMC article.
-
Knockdown of CLC-3 in the hippocampal CA1 impairs contextual fear memory.Prog Neuropsychopharmacol Biol Psychiatry. 2019 Mar 8;89:132-145. doi: 10.1016/j.pnpbp.2018.07.004. Epub 2018 Jul 17. Prog Neuropsychopharmacol Biol Psychiatry. 2019. PMID: 30025794
-
Ca2+/calmodulin-dependent protein kinase II and protein kinase C activities mediate extracellular glucose-regulated hippocampal synaptic efficacy.Mol Cell Neurosci. 2011 Jan;46(1):101-7. doi: 10.1016/j.mcn.2010.08.010. Epub 2010 Aug 31. Mol Cell Neurosci. 2011. PMID: 20807573
-
Latent Sex Differences in Molecular Signaling That Underlies Excitatory Synaptic Potentiation in the Hippocampus.J Neurosci. 2019 Feb 27;39(9):1552-1565. doi: 10.1523/JNEUROSCI.1897-18.2018. Epub 2018 Dec 21. J Neurosci. 2019. PMID: 30578341 Free PMC article.
-
CaM kinase II in long-term potentiation.Neurochem Int. 1996 Apr;28(4):343-58. doi: 10.1016/0197-0186(95)00097-6. Neurochem Int. 1996. PMID: 8740440 Review.
Cited by
-
Modulation of GABAergic transmission in development and neurodevelopmental disorders: investigating physiology and pathology to gain therapeutic perspectives.Front Cell Neurosci. 2014 May 22;8:119. doi: 10.3389/fncel.2014.00119. eCollection 2014. Front Cell Neurosci. 2014. PMID: 24904277 Free PMC article. Review.
-
Chloride Homeostasis in Neurons With Special Emphasis on the Olivocerebellar System: Differential Roles for Transporters and Channels.Front Cell Neurosci. 2018 May 1;12:101. doi: 10.3389/fncel.2018.00101. eCollection 2018. Front Cell Neurosci. 2018. PMID: 29765304 Free PMC article. Review.
-
ClC-3 Expression and Its Association with Hyperglycemia Induced HT22 Hippocampal Neuronal Cell Apoptosis.J Diabetes Res. 2016;2016:2984380. doi: 10.1155/2016/2984380. Epub 2016 Jan 26. J Diabetes Res. 2016. PMID: 26925421 Free PMC article.
-
Neuronal ClC-3 Splice Variants Differ in Subcellular Localizations, but Mediate Identical Transport Functions.J Biol Chem. 2015 Oct 23;290(43):25851-62. doi: 10.1074/jbc.M115.668186. Epub 2015 Sep 4. J Biol Chem. 2015. PMID: 26342074 Free PMC article.
-
Chloride channel-3 promotes tumor metastasis by regulating membrane ruffling and is associated with poor survival.Oncotarget. 2015 Feb 10;6(4):2434-50. doi: 10.18632/oncotarget.2966. Oncotarget. 2015. PMID: 25537517 Free PMC article.
References
-
- Albensi BC, Oliver DR, Toupin J, Odero G. Electrical stimulation protocols for hippocampal synaptic plasticity and neuronal hyper-excitability: are they effective or relevant. Exp Neurol. 2007;204:1–13. - PubMed
-
- Bats C, Groc L, Choquet D. The interaction between Stargazin and PSD-95 regulates AMPA receptor surface trafficking. Neuron. 2007;53:719–734. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

