TCPTP regulates SFK and STAT3 signaling and is lost in triple-negative breast cancers
- PMID: 23166300
- PMCID: PMC3554209
- DOI: 10.1128/MCB.01016-12
TCPTP regulates SFK and STAT3 signaling and is lost in triple-negative breast cancers
Erratum in
-
Correction.Mol Cell Biol. 2023;43(11):629. doi: 10.1080/10985549.2023.2277100. Epub 2023 Nov 17. Mol Cell Biol. 2023. PMID: 37955527 Free PMC article. No abstract available.
Abstract
Tyrosine phosphorylation-dependent signaling, as mediated by members of the epidermal growth factor receptor (EGFR) family (ErbB1 to -4) of protein tyrosine kinases (PTKs), Src family PTKs (SFKs), and cytokines such as interleukin-6 (IL-6) that signal via signal transducer and activator of transcription 3 (STAT3), is critical to the development and progression of many human breast cancers. EGFR, SFKs, and STAT3 can serve as substrates for the protein tyrosine phosphatase TCPTP (PTPN2). Here we report that TCPTP protein levels are decreased in a subset of breast cancer cell lines in vitro and that TCPTP protein is absent in a large proportion of "triple-negative" primary human breast cancers. Homozygous TCPTP deficiency in murine mammary fat pads in vivo is associated with elevated SFK and STAT3 signaling, whereas TCPTP deficiency in human breast cancer cell lines enhances SFK and STAT3 signaling. On the other hand, TCPTP reconstitution in human breast cancer cell lines severely impaired cell proliferation and suppressed anchorage-independent growth in vitro and xenograft growth in vivo. These studies establish TCPTP's potential to serve as a tumor suppressor in human breast cancer.
Figures


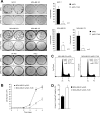




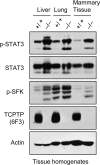

Comment in
-
On the role of tyrosine phosphatases as negative regulators of STAT signaling in breast cancers: new findings and future perspectives.Breast Cancer Res. 2013 Jul 31;15(4):312. doi: 10.1186/bcr3437. Breast Cancer Res. 2013. PMID: 23905670 Free PMC article.
References
-
- Yarden Y. 2001. Biology of HER2 and its importance in breast cancer. Oncology 61(Suppl 2):1–13 - PubMed
-
- Nielsen TO, Hsu FD, Jensen K, Cheang M, Karaca G, Hu Z, Hernandez-Boussard T, Livasy C, Cowan D, Dressler L, Akslen LA, Ragaz J, Gown AM, Gilks CB, van de Rijn M, Perou CM. 2004. Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin. Cancer Res. 10:5367–5374 - PubMed
-
- Biscardi JS, Belsches AP, Parsons SJ. 1998. Characterization of human epidermal growth factor receptor and c-Src interactions in human breast tumor cells. Mol. Carcinog. 21:261–272 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
