The contributions of wobbling and superwobbling to the reading of the genetic code
- PMID: 23166520
- PMCID: PMC3499367
- DOI: 10.1371/journal.pgen.1003076
The contributions of wobbling and superwobbling to the reading of the genetic code
Abstract
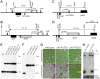
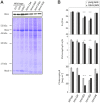
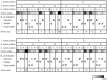
Reduced bacterial genomes and most genomes of cell organelles (chloroplasts and mitochondria) do not encode the full set of 32 tRNA species required to read all triplets of the genetic code according to the conventional wobble rules. Superwobbling, in which a single tRNA species that contains a uridine in the wobble position of the anticodon reads an entire four-fold degenerate codon box, has been suggested as a possible mechanism for how tRNA sets can be reduced. However, the general feasibility of superwobbling and its efficiency in the various codon boxes have remained unknown. Here we report a complete experimental assessment of the decoding rules in a typical prokaryotic genetic system, the plastid genome. By constructing a large set of transplastomic knock-out mutants for pairs of isoaccepting tRNA species, we show that superwobbling occurs in all codon boxes where it is theoretically possible. Phenotypic characterization of the transplastomic mutant plants revealed that the efficiency of superwobbling varies in a codon box-dependent manner, but--contrary to previous suggestions--it is independent of the number of hydrogen bonds engaged in codon-anticodon interaction. Finally, our data provide experimental evidence of the minimum tRNA set comprising 25 tRNA species, a number lower than previously suggested. Our results demonstrate that all triplets with pyrimidines in third codon position are dually decoded: by a tRNA species utilizing standard base pairing or wobbling and by a second tRNA species employing superwobbling. This has important implications for the interpretation of the genetic code and will aid the construction of synthetic genomes with a minimum-size translational apparatus.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




References
-
- Crick FHC (1966) Codon-anticodon pairing: The wobble hypothesis. J Mol Biol 19: 548–555. - PubMed
-
- Schneider A (1994) Import of RNA into mitochondria. Trends Cell Biol 4: 282–286. - PubMed
-
- Duchêne A-M, Pujol C, Maréchal-Drouard L (2009) Import of tRNAs and aminoacyl-tRNA synthetases into mitochondria. Curr Genet 55: 1–18. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

