Histone methyltransferases MES-4 and MET-1 promote meiotic checkpoint activation in Caenorhabditis elegans
- PMID: 23166523
- PMCID: PMC3499413
- DOI: 10.1371/journal.pgen.1003089
Histone methyltransferases MES-4 and MET-1 promote meiotic checkpoint activation in Caenorhabditis elegans
Abstract
Chromosomes that fail to synapse during meiosis become enriched for chromatin marks associated with heterochromatin assembly. This response, called meiotic silencing of unsynapsed or unpaired chromatin (MSUC), is conserved from fungi to mammals. In Caenorhabditis elegans, unsynapsed chromosomes also activate a meiotic checkpoint that monitors synapsis. The synapsis checkpoint signal is dependent on cis-acting loci called Pairing Centers (PCs). How PCs signal to activate the synapsis checkpoint is currently unknown. We show that a chromosomal duplication with PC activity is sufficient to activate the synapsis checkpoint and that it undergoes heterochromatin assembly less readily than a duplication of a non-PC region, suggesting that the chromatin state of these loci is important for checkpoint function. Consistent with this hypothesis, MES-4 and MET-1, chromatin-modifying enzymes associated with transcriptional activity, are required for the synapsis checkpoint. In addition, a duplication with PC activity undergoes heterochromatin assembly when mes-4 activity is reduced. MES-4 function is required specifically for the X chromosome, while MES-4 and MET-1 act redundantly to monitor autosomal synapsis. We propose that MES-4 and MET-1 antagonize heterochromatin assembly at PCs of unsynapsed chromosomes by promoting a transcriptionally permissive chromatin environment that is required for meiotic checkpoint function. Moreover, we suggest that different genetic requirements to monitor the behavior of sex chromosomes and autosomes allow for the lone unsynapsed X present in male germlines to be shielded from inappropriate checkpoint activation.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
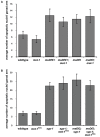
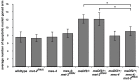
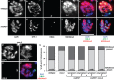
Similar articles
-
Caenorhabditis elegans histone methyltransferase MET-2 shields the male X chromosome from checkpoint machinery and mediates meiotic sex chromosome inactivation.PLoS Genet. 2011 Sep;7(9):e1002267. doi: 10.1371/journal.pgen.1002267. Epub 2011 Sep 1. PLoS Genet. 2011. PMID: 21909284 Free PMC article.
-
Plasticity in the Meiotic Epigenetic Landscape of Sex Chromosomes in Caenorhabditis Species.Genetics. 2016 Aug;203(4):1641-58. doi: 10.1534/genetics.116.191130. Epub 2016 Jun 8. Genetics. 2016. PMID: 27280692 Free PMC article.
-
Synaptonemal Complex Components Are Required for Meiotic Checkpoint Function in Caenorhabditis elegans.Genetics. 2016 Nov;204(3):987-997. doi: 10.1534/genetics.116.191494. Epub 2016 Sep 7. Genetics. 2016. PMID: 27605049 Free PMC article.
-
Meiotic silencing in Caenorhabditis elegans.Int Rev Cell Mol Biol. 2010;282:91-134. doi: 10.1016/S1937-6448(10)82002-7. Epub 2010 Jun 18. Int Rev Cell Mol Biol. 2010. PMID: 20630467 Review.
-
DNA double strand break repair, chromosome synapsis and transcriptional silencing in meiosis.Epigenetics. 2010 May 16;5(4):255-66. doi: 10.4161/epi.5.4.11518. Epub 2010 May 16. Epigenetics. 2010. PMID: 20364103 Review.
Cited by
-
Evidence that masking of synapsis imperfections counterbalances quality control to promote efficient meiosis.PLoS Genet. 2013;9(12):e1003963. doi: 10.1371/journal.pgen.1003963. Epub 2013 Dec 5. PLoS Genet. 2013. PMID: 24339786 Free PMC article.
-
Histone methylation restrains the expression of subtype-specific genes during terminal neuronal differentiation in Caenorhabditis elegans.PLoS Genet. 2013;9(12):e1004017. doi: 10.1371/journal.pgen.1004017. Epub 2013 Dec 12. PLoS Genet. 2013. PMID: 24348272 Free PMC article.
-
Bisection of the X chromosome disrupts the initiation of chromosome silencing during meiosis in Caenorhabditis elegans.Nat Commun. 2021 Aug 10;12(1):4802. doi: 10.1038/s41467-021-24815-0. Nat Commun. 2021. PMID: 34376665 Free PMC article.
-
Needhi Bhalla: Chromosomes do the most amazing things.J Cell Biol. 2016 Feb 1;212(3):260-1. doi: 10.1083/jcb.2123pi. J Cell Biol. 2016. PMID: 26833784 Free PMC article.
-
CDK2 regulates the NRF1/Ehmt1 axis during meiotic prophase I.J Cell Biol. 2019 Sep 2;218(9):2896-2918. doi: 10.1083/jcb.201903125. Epub 2019 Jul 26. J Cell Biol. 2019. PMID: 31350280 Free PMC article.
References
-
- Hassold T, Hunt P (2001) To err (meiotically) is human: the genesis of human aneuploidy. Nat Rev Genet 2: 280–291. - PubMed
-
- MacQueen AJ, Hochwagen A (2011) Checkpoint mechanisms: the puppet masters of meiotic prophase. Trends in cell biology 21: 393–400. - PubMed
-
- Bhalla N, Dernburg AF (2005) A conserved checkpoint monitors meiotic chromosome synapsis in Caenorhabditis elegans. Science 310: 1683–1686. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

