The co-chaperone Hch1 regulates Hsp90 function differently than its homologue Aha1 and confers sensitivity to yeast to the Hsp90 inhibitor NVP-AUY922
- PMID: 23166640
- PMCID: PMC3498168
- DOI: 10.1371/journal.pone.0049322
The co-chaperone Hch1 regulates Hsp90 function differently than its homologue Aha1 and confers sensitivity to yeast to the Hsp90 inhibitor NVP-AUY922
Abstract
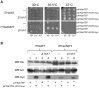
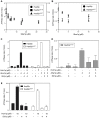
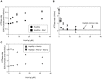
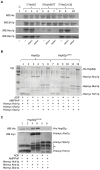
Hsp90 is a dimeric ATPase responsible for the activation or maturation of a specific set of substrate proteins termed 'clients'. This molecular chaperone acts in the context of a structurally dynamic and highly regulated cycle involving ATP, co-chaperone proteins and clients. Co-chaperone proteins regulate conformational transitions that may be impaired in mutant forms of Hsp90. We report here that the in vivo impairment of commonly studied Hsp90 variants harbouring the G313S or A587T mutation are exacerbated by the co-chaperone Hch1p. Deletion of HCH1, but not AHA1, mitigates the temperature sensitive phenotype and high sensitivity to Hsp90 inhibitor drugs observed in Saccharomyces cerevisiae that express either of these two Hsp90 variants. Moreover, the deletion of HCH1 results in high resistance to Hsp90 inhibitors in yeast that express wildtype Hsp90. Conversely, the overexpression of Hch1p greatly increases sensitivity to Hsp90 inhibition in yeast expressing wildtype Hsp90. We conclude that despite the similarity between these two co-chaperones, Hch1p and Aha1p regulate Hsp90 function in distinct ways and likely independent of their roles as ATPase stimulators. We further conclude that Hch1p plays a critical role in regulating Hsp90 inhibitor drug sensitivity in yeast.
Conflict of interest statement
Figures




Similar articles
-
A mutation in the catalytic loop of Hsp90 specifically impairs ATPase stimulation by Aha1p, but not Hch1p.J Mol Biol. 2014 Jun 12;426(12):2379-92. doi: 10.1016/j.jmb.2014.04.002. Epub 2014 Apr 12. J Mol Biol. 2014. PMID: 24726918
-
Aha1 binds to the middle domain of Hsp90, contributes to client protein activation, and stimulates the ATPase activity of the molecular chaperone.J Biol Chem. 2003 May 9;278(19):17228-35. doi: 10.1074/jbc.M212761200. Epub 2003 Feb 24. J Biol Chem. 2003. PMID: 12604615
-
Integration of the accelerator Aha1 in the Hsp90 co-chaperone cycle.Nat Struct Mol Biol. 2013 Mar;20(3):326-31. doi: 10.1038/nsmb.2502. Epub 2013 Feb 10. Nat Struct Mol Biol. 2013. PMID: 23396352
-
p23 and Aha1: Distinct Functions Promote Client Maturation.Subcell Biochem. 2023;101:159-187. doi: 10.1007/978-3-031-14740-1_6. Subcell Biochem. 2023. PMID: 36520307 Review.
-
Aha-type co-chaperones: the alpha or the omega of the Hsp90 ATPase cycle?Biol Chem. 2020 Mar 26;401(4):423-434. doi: 10.1515/hsz-2019-0341. Biol Chem. 2020. PMID: 31782942 Review.
Cited by
-
p53 protein regulates Hsp90 ATPase activity and thereby Wnt signaling by modulating Aha1 expression.J Biol Chem. 2014 Mar 7;289(10):6513-6525. doi: 10.1074/jbc.M113.532523. Epub 2014 Jan 22. J Biol Chem. 2014. Retraction in: J Biol Chem. 2020 Jan 3;295(1):289. doi: 10.1074/jbc.W119.012134. PMID: 24451373 Free PMC article. Retracted.
-
The Mechanism of Hsp90 ATPase Stimulation by Aha1.Sci Rep. 2016 Sep 12;6:33179. doi: 10.1038/srep33179. Sci Rep. 2016. PMID: 27615124 Free PMC article.
-
Mutation of essential Hsp90 co-chaperones SGT1 or CNS1 renders yeast hypersensitive to overexpression of other co-chaperones.Curr Genet. 2014 Nov;60(4):265-76. doi: 10.1007/s00294-014-0432-3. Epub 2014 Jun 13. Curr Genet. 2014. PMID: 24923785
-
Evidence for interaction between Hsp90 and the ER membrane complex.Cell Stress Chaperones. 2018 Sep;23(5):1101-1115. doi: 10.1007/s12192-018-0908-z. Epub 2018 May 28. Cell Stress Chaperones. 2018. PMID: 29808299 Free PMC article.
-
Collaboration between two conserved sequence motifs drives ATPase stimulation of Hsp90 by Aha1.bioRxiv [Preprint]. 2025 Jun 23:2025.06.10.658861. doi: 10.1101/2025.06.10.658861. bioRxiv. 2025. PMID: 40667101 Free PMC article. Preprint.
References
-
- Taipale M, Jarosz DF, Lindquist S (2010) HSP90 at the hub of protein homeostasis: emerging mechanistic insights. Nat Rev Mol Cell Biol 11: 515–528. - PubMed
-
- Ansar S, Burlison JA, Hadden MK, Yu XM, Desino KE, et al. (2007) A non-toxic Hsp90 inhibitor protects neurons from Abeta-induced toxicity. Bioorg Med Chem Lett 17: 1984–1990. - PubMed
-
- Barral JM, Broadley SA, Schaffar G, Hartl FU (2004) Roles of molecular chaperones in protein misfolding diseases. Semin Cell Dev Biol 15: 17–29. - PubMed
-
- Citri A, Kochupurakkal BS, Yarden Y (2004) The achilles heel of ErbB-2/HER2: regulation by the Hsp90 chaperone machine and potential for pharmacological intervention. Cell Cycle 3: 51–60. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

