Altered nitric oxide production mediates matrix-specific PAK2 and NF-κB activation by flow
- PMID: 23171552
- PMCID: PMC3564533
- DOI: 10.1091/mbc.E12-07-0513
Altered nitric oxide production mediates matrix-specific PAK2 and NF-κB activation by flow
Abstract
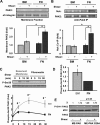
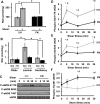
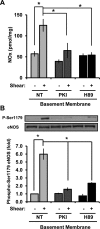
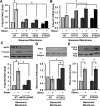
Shear stress generated by distinct blood flow patterns modulates endothelial cell phenotype to spatially restrict atherosclerotic plaque development. Signaling through p21-activated kinase (PAK) mediates several of the deleterious effects of shear stress, including enhanced NF-κB activation and proinflammatory gene expression. Whereas shear stress activates PAK in endothelial cells on a fibronectin matrix, basement membrane proteins limit shear-induced PAK activation and inflammation through a protein kinase A-dependent pathway; however, the mechanisms underlying this regulation were unknown. We show that basement membrane proteins limit membrane recruitment of PAK2, the dominant isoform in endothelial cells, by blocking its interaction with the adaptor protein Nck. This uncoupling response requires protein kinase A-dependent nitric oxide production and subsequent PAK2 phosphorylation on Ser-20 in the Nck-binding domain. Of importance, shear stress does not stimulate nitric oxide production in endothelial cells on fibronectin, resulting in enhanced PAK activation, NF-κB phosphorylation, ICAM-1 expression, and monocyte adhesion. These data demonstrate that differential flow-induced nitric oxide production regulates matrix-specific PAK signaling and describe a novel mechanism of nitric oxide-dependent NF-κB inhibition.
Figures


Similar articles
-
Matrix-specific protein kinase A signaling regulates p21-activated kinase activation by flow in endothelial cells.Circ Res. 2010 Apr 30;106(8):1394-403. doi: 10.1161/CIRCRESAHA.109.210286. Epub 2010 Mar 11. Circ Res. 2010. PMID: 20224042 Free PMC article.
-
Nck1, But Not Nck2, Mediates Disturbed Flow-Induced p21-Activated Kinase Activation and Endothelial Permeability.J Am Heart Assoc. 2020 Jun 2;9(11):e016099. doi: 10.1161/JAHA.120.016099. Epub 2020 May 29. J Am Heart Assoc. 2020. PMID: 32468886 Free PMC article.
-
p21-activated kinase signaling regulates oxidant-dependent NF-kappa B activation by flow.Circ Res. 2008 Sep 12;103(6):671-9. doi: 10.1161/CIRCRESAHA.108.182097. Epub 2008 Jul 31. Circ Res. 2008. PMID: 18669917 Free PMC article.
-
Regulation of Vascular Injury and Repair by P21-Activated Kinase 1 and P21-Activated Kinase 2: Therapeutic Potential and Challenges.Biomolecules. 2024 Dec 13;14(12):1596. doi: 10.3390/biom14121596. Biomolecules. 2024. PMID: 39766303 Free PMC article. Review.
-
Shear stress and plaque development.Expert Rev Cardiovasc Ther. 2010 Apr;8(4):545-56. doi: 10.1586/erc.10.28. Expert Rev Cardiovasc Ther. 2010. PMID: 20397828 Free PMC article. Review.
Cited by
-
Inhibiting Integrin α5 Cytoplasmic Domain Signaling Reduces Atherosclerosis and Promotes Arteriogenesis.J Am Heart Assoc. 2018 Jan 30;7(3):e007501. doi: 10.1161/JAHA.117.007501. J Am Heart Assoc. 2018. PMID: 29382667 Free PMC article.
-
Biochemical and mechanical signals in the lymphatic vasculature.Cell Mol Life Sci. 2021 Aug;78(16):5903-5923. doi: 10.1007/s00018-021-03886-8. Epub 2021 Jul 8. Cell Mol Life Sci. 2021. PMID: 34240226 Free PMC article. Review.
-
p21-Activated Kinase 2 Regulates Endothelial Development and Function through the Bmk1/Erk5 Pathway.Mol Cell Biol. 2015 Dec;35(23):3990-4005. doi: 10.1128/MCB.00630-15. Epub 2015 Sep 21. Mol Cell Biol. 2015. PMID: 26391956 Free PMC article.
-
cSTAR analysis identifies endothelial cell cycle as a key regulator of flow-dependent artery remodeling.Sci Adv. 2025 Jan 3;11(1):eado9970. doi: 10.1126/sciadv.ado9970. Epub 2025 Jan 3. Sci Adv. 2025. PMID: 39752487 Free PMC article.
-
Sinner or Saint?: Nck Adaptor Proteins in Vascular Biology.Front Cell Dev Biol. 2021 May 26;9:688388. doi: 10.3389/fcell.2021.688388. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34124074 Free PMC article. Review.
References
-
- Bokoch GM. Biology of the p21-activated kinases. Annu Rev Biochem. 2003;72:743–781. - PubMed
-
- Boo YC, Hwang J, Sykes M, Michell BJ, Kemp BE, Lum H, Jo H. Shear stress stimulates phosphorylation of eNOS at Ser(635) by a protein kinase A-dependent mechanism. Am J Physiol Heart Circ Physiol. 2002a;283:H1819–H1828. - PubMed
-
- Boo YC, Sorescu G, Boyd N, Shiojima I, Walsh K, Du J, Jo H. Shear stress stimulates phosphorylation of endothelial nitric-oxide synthase at Ser1179 by Akt-independent mechanisms: role of protein kinase A. J Biol Chem. 2002b;277:3388–3396. - PubMed
-
- Boo YC, Sorescu GP, Bauer PM, Fulton D, Kemp BE, Harrison DG, Sessa WC, Jo H. Endothelial NO synthase phosphorylated at SER635 produces NO without requiring intracellular calcium increase. Free Radic Biol Med. 2003;35:729–741. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

