Mitochondria "fuel" breast cancer metabolism: fifteen markers of mitochondrial biogenesis label epithelial cancer cells, but are excluded from adjacent stromal cells
- PMID: 23172368
- PMCID: PMC3552922
- DOI: 10.4161/cc.22777
Mitochondria "fuel" breast cancer metabolism: fifteen markers of mitochondrial biogenesis label epithelial cancer cells, but are excluded from adjacent stromal cells
Abstract
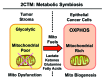
Here, we present new genetic and morphological evidence that human tumors consist of two distinct metabolic compartments. First, re-analysis of genome-wide transcriptional profiling data revealed that > 95 gene transcripts associated with mitochondrial biogenesis and/or mitochondrial translation were significantly elevated in human breast cancer cells, as compared with adjacent stromal tissue. Remarkably, nearly 40 of these upregulated gene transcripts were mitochondrial ribosomal proteins (MRPs), functionally associated with mitochondrial translation of protein components of the OXPHOS complex. Second, during validation by immunohistochemistry, we observed that antibodies directed against 15 markers of mitochondrial biogenesis and/or mitochondrial translation (AKAP1, GOLPH3, GOLPH3L, MCT1, MRPL40, MRPS7, MRPS15, MRPS22, NRF1, NRF2, PGC1-α, POLRMT, TFAM, TIMM9 and TOMM70A) selectively labeled epithelial breast cancer cells. These same mitochondrial markers were largely absent or excluded from adjacent tumor stromal cells. Finally, markers of mitochondrial lipid synthesis (GOLPH3) and mitochondrial translation (POLRMT) were associated with poor clinical outcome in human breast cancer patients. Thus, we conclude that human breast cancers contain two distinct metabolic compartments-a glycolytic tumor stroma, which surrounds oxidative epithelial cancer cells-that are mitochondria-rich. The co-existence of these two compartments is indicative of metabolic symbiosis between epithelial cancer cells and their surrounding stroma. As such, epithelial breast cancer cells should be viewed as predatory metabolic "parasites," which undergo anabolic reprogramming to amplify their mitochondrial "power." This notion is consistent with the observation that the anti-malarial agent chloroquine may be an effective anticancer agent. New anticancer therapies should be developed to target mitochondrial biogenesis and/or mitochondrial translation in human cancer cells.
Figures







References
-
- Pavlides S, Vera I, Gandara R, Sneddon S, Pestell RG, Mercier I, et al. Warburg meets autophagy: cancer-associated fibroblasts accelerate tumor growth and metastasis via oxidative stress, mitophagy, and aerobic glycolysis. Antioxid Redox Signal. 2012;16:1264–84. doi: 10.1089/ars.2011.4243. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
