Novel route to chaetomellic acid A and analogues: serendipitous discovery of a more competent FTase inhibitor
- PMID: 23182215
- PMCID: PMC3761967
- DOI: 10.1016/j.bmc.2012.10.034
Novel route to chaetomellic acid A and analogues: serendipitous discovery of a more competent FTase inhibitor
Abstract
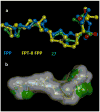
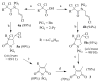
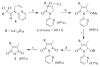
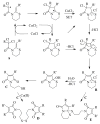
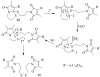
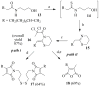
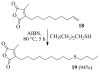
A new practical route to chaetomellic acid A (ACA), based on the copper catalysed radical cyclization (RC) of (Z)-3-(2,2-dichloropropanoyl)-2-pentadecylidene-1,3-thiazinane, is described. Remarkably, the process entailed: (i) a one-pot preparation of the intermediate N-α-perchloroacyl-2-(Z)-alkyliden-1,3-thiazinanes starting from N-(3-hydroxypropyl)palmitamide, (ii) a two step smooth transformation of the RC products into ACA and (iii) only one intermediate chromatographic purification step. The method offers a versatile approach to the preparation of ACA analogues, through the synthesis of an intermediate maleic anhydride with a vinylic group at the end of the aliphatic tail, a function that can be transformed through a thiol-ene coupling. Serendipitously, the disodium salt of 2-(9-(butylthio)nonyl)-3-methylmaleic acid, that we prepared as a representative sulfurated ACA analogue, was a more competent FTase inhibitor than ACA. This behaviour was analysed by a molecular docking study.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures
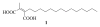

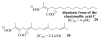

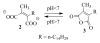
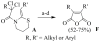
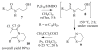

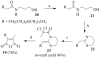
Similar articles
-
Efficient syntheses, human and yeast farnesyl-protein transferase inhibitory activities of chaetomellic acids and analogues.Bioorg Med Chem. 2000 Mar;8(3):571-80. doi: 10.1016/s0968-0896(99)00312-0. Bioorg Med Chem. 2000. PMID: 10732974
-
A simple and efficient synthesis of the ras farnesyl-protein transferase inhibitor chaetomellic acid A.Bioorg Med Chem. 1996 Jun;4(6):881-3. doi: 10.1016/0968-0896(96)00076-4. Bioorg Med Chem. 1996. PMID: 8818238
-
Theoretical studies on binding and specificity mechanisms of farnesyltransferase (FTase) and geranylgeranyltransferase type-I (GGTase-I) inhibitors by molecular modeling.Comb Chem High Throughput Screen. 2014;17(6):509-19. doi: 10.2174/1386207317666140120122920. Comb Chem High Throughput Screen. 2014. PMID: 24444014
-
Farnesyltransferase inhibitors: a detailed chemical view on an elusive biological problem.Curr Med Chem. 2008;15(15):1478-92. doi: 10.2174/092986708784638825. Curr Med Chem. 2008. PMID: 18537624 Review.
-
Natural products with maleic anhydride structure: nonadrides, tautomycin, chaetomellic anhydride, and other compounds.Chem Rev. 2007 May;107(5):1777-830. doi: 10.1021/cr050029r. Epub 2007 Apr 18. Chem Rev. 2007. PMID: 17439289 Review. No abstract available.
Cited by
-
Targeting prenylation inhibition through the mevalonate pathway.RSC Med Chem. 2019 Dec 23;11(1):51-71. doi: 10.1039/c9md00442d. eCollection 2020 Jan 1. RSC Med Chem. 2019. PMID: 33479604 Free PMC article. Review.
-
New tricks for human farnesyltransferase inhibitor: cancer and beyond.Medchemcomm. 2017 Feb 16;8(5):841-854. doi: 10.1039/c7md00030h. eCollection 2017 May 1. Medchemcomm. 2017. PMID: 30108801 Free PMC article. Review.
-
Chemistry of Substituted Thiazinanes and Their Derivatives.Molecules. 2020 Nov 28;25(23):5610. doi: 10.3390/molecules25235610. Molecules. 2020. PMID: 33260625 Free PMC article. Review.
-
Bicyclic N,S-Acetals Containing Fused Cysteine-Amide System as New Heterocyclic Class Targeting Human Farnesyltransferase (FTase-h).Int J Mol Sci. 2025 Feb 17;26(4):1717. doi: 10.3390/ijms26041717. Int J Mol Sci. 2025. PMID: 40004178 Free PMC article.
References
-
- Leonard DM. J Med Chem. 1997;40:2971–2990. - PubMed
- Beck LA, Hosick TJ, Sinensky M. J Cell Biol. 1988;107:1307–1316. - PMC - PubMed
- Wolda SL, Glomset JA. J Biol Chem. 1988;263:5997–6000. - PubMed
- Lane KT, Beese LS. J Lipid Res. 2006;47:681–699. - PubMed
- Winter-Vann AM, Casey PJ. Nat Rev Cancer. 2005;5:405–412. - PubMed
-
- Anderegg RJ, Betz R, Carr SA, Crabb JW, Duntze W. J Biol Chem. 1988;263:18236–18240. - PubMed
- Farnsworth CC, Wolda CL, Gelb MH, Glomset JA. J Biol Chem. 1989;264:20422–20429. - PMC - PubMed
- Casey PJ, Solski PA, Der CJ, Buss JE. Proc Natl Acad Sci USA. 1989;86:8323–8327. - PMC - PubMed
- Maltese WA, Robishaw JD. J Biol Chem. 1990;265:18071–18074. - PubMed
- Fukada Y, Takao T, Ohguru H, Yoshizawa T, Akino T, Shimonishi Y. Nature (London) 1990;346:658–660. - PubMed
- Lai RK, Perez-Sala D, Canada FJ, Rando RR. Proc Natl Acad Sci USA. 1990;87:7673–7677. - PMC - PubMed
- Moores SL, Schaber MD, Mosser SD, Rands E, O'Hara MB, Garsky VM, Marshall MS, Pompliano DL, Gibbs JB. J Biol Chem. 1991;266:14603–14610. - PubMed
-
- Chow M, Der CJ, Buss JE. Curr Opinion Cell Biol. 1992;4:629–636. - PubMed
-
- Schafer WR, Rine J. Annu Rev Genet. 1992;26:209–237. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

