Cardiovascular safety of anagrelide in healthy subjects: effects of caffeine and food intake on pharmacokinetics and adverse reactions
- PMID: 23184666
- PMCID: PMC3586167
- DOI: 10.1007/s40261-012-0032-2
Cardiovascular safety of anagrelide in healthy subjects: effects of caffeine and food intake on pharmacokinetics and adverse reactions
Abstract
Background: Essential thrombocythaemia (ET) is a rare clonal myeloproliferative disorder characterized by a sustained elevation in platelet count and megakaryocyte hyperplasia. Anagrelide is used in the treatment of ET, where it has been shown to reduce platelet count. Anagrelide is metabolized by cytochrome P450 (CYP) 1A2, and previous studies of the effect of food on the bioavailability and pharmacokinetics of anagrelide were conducted prior to the identification of the active metabolite, 3-hydroxyanagrelide.
Objectives: The objectives of this study were to determine the effect of food and caffeine on the pharmacokinetics of anagrelide and its active metabolite, 3-hydroxyanagrelide, to monitor electrocardiogram (ECG) parameters following drug administration, and to document the relationship between palpitations, ECG changes and caffeine intake
Methods: Thirty-five healthy subjects who received 1 mg of anagrelide following either a 10-h fast or within 30 min of a standardized breakfast, including two cups of coffee, were studied.
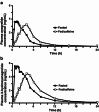
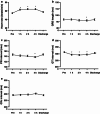
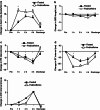
Results: Time to maximum (peak) plasma concentration (C(max)) of anagrelide was 4.0 h in the fed and 1.5 h in the fasted group (p < 0.05); similar results were observed for 3-hydroxyanagrelide. The mean C(max) of anagrelide was 4.45 ± 2.32 ng/mL and 5.08 ± 2.99 ng/mL in the fed/caffeine and fasted groups, respectively; peak concentrations were higher for 3-hydroxyanagrelide in both the fed/caffeine and fasted groups. The most frequent adverse events (AEs) were headache (60 %) and palpitations (40 %). There were no serious AEs and all ECGs were normal, although significant reductions in PR interval, QRS length and QT interval were observed in both groups. Heart rate increased after anagrelide administration in both fed/caffeine and fasted states (p < 0.01); however, increased heart rate was significantly more frequent in the fed/caffeine state than in the fasted state (p < 0.001 for heart rate increase in the first hour after drug administration). There was a trend towards a greater heart rate increase in subjects reporting palpitations than in those without (mean heart rate ± SD at 1 h: 10.1 ± 6.4 vs. 8.0 ± 8.4 beats/min [p = 0.35]; at 4 h: 12.7 ± 7.5 vs. 9.1 ± 8.8 beats/min [p = 0.10], respectively).
Conclusion: We conclude that food/caffeine delayed absorption of anagrelide. Anagrelide was generally well tolerated and had small effects on ECG parameters and heart rate. Caffeine may be implicated in a higher increase in heart rate and increased frequency of palpitations observed following administration of anagrelide with food/caffeine versus fasting.
Figures
Similar articles
-
Pharmacokinetics and tolerability of anagrelide hydrochloride in young (18 - 50 years) and elderly (≥ 65 years) patients with essential thrombocythemia.Int J Clin Pharmacol Ther. 2012 Nov;50(11):787-96. doi: 10.5414/CP201711. Int J Clin Pharmacol Ther. 2012. PMID: 22943924 Clinical Trial.
-
Pharmacokinetics, bioequivalence, tolerability, and effects on platelet counts of two formulations of anagrelide in healthy volunteers and patients with thrombocythemia associated with chronic myeloproliferation.Clin Ther. 2009 Feb;31(2):386-98. doi: 10.1016/j.clinthera.2009.02.008. Clin Ther. 2009. PMID: 19302911 Clinical Trial.
-
Pharmacokinetics of vandetanib: three phase I studies in healthy subjects.Clin Ther. 2012 Jan;34(1):221-37. doi: 10.1016/j.clinthera.2011.11.011. Epub 2011 Dec 28. Clin Ther. 2012. PMID: 22206795 Clinical Trial.
-
Anagrelide, a selective thrombocytopenic agent.Am J Health Syst Pharm. 1998 Oct 1;55(19):1979-86. doi: 10.1093/ajhp/55.19.1979. Am J Health Syst Pharm. 1998. PMID: 9784784 Review.
-
Anagrelide: a novel agent for the treatment of myeloproliferative disorders.Expert Opin Pharmacother. 2000 Mar;1(3):537-46. doi: 10.1517/14656566.1.3.537. Expert Opin Pharmacother. 2000. PMID: 11249536 Review.
Cited by
-
Hybrid System for Engagement Recognition During Cognitive Tasks Using a CFS + KNN Algorithm.Sensors (Basel). 2018 Oct 30;18(11):3691. doi: 10.3390/s18113691. Sensors (Basel). 2018. PMID: 30380784 Free PMC article.
-
In Vitro and In Vivo Studies of Melanoma Cell Migration by Antagonistic Mimetics of Adhesion Molecule L1CAM.Int J Mol Sci. 2024 Apr 28;25(9):4811. doi: 10.3390/ijms25094811. Int J Mol Sci. 2024. PMID: 38732030 Free PMC article.
-
Pharmacokinetics of a Novel Anagrelide Extended-Release Formulation in Healthy Subjects: Food Intake and Comparison With a Reference Product.Clin Pharmacol Drug Dev. 2018 Feb;7(2):123-131. doi: 10.1002/cpdd.340. Epub 2017 Mar 16. Clin Pharmacol Drug Dev. 2018. PMID: 28301098 Free PMC article. Clinical Trial.
-
Pharmacokinetic profile and in vivo anticancer efficacy of anagrelide administered subcutaneously in rodents.Drug Deliv. 2025 Dec;32(1):2463433. doi: 10.1080/10717544.2025.2463433. Epub 2025 Feb 10. Drug Deliv. 2025. PMID: 39930717 Free PMC article.
-
Ventricular tachyarrhythmia in a 78-year-old woman with essential thrombocythaemia.BMJ Case Rep. 2018 Feb 8;2018:bcr2017220723. doi: 10.1136/bcr-2017-220723. BMJ Case Rep. 2018. PMID: 29437800 Free PMC article.
References
-
- Failkow PJ, Faguet GB, Jacobson RJ, et al. Evidence that essential thrombocythemia is a clonal disorder with origin in a multipotent stem cell. Blood. 1981;58:916–919. - PubMed
-
- Birgegård G, Björkholm M, Kutti J, et al. Adverse effects and benefits of two years of anagrelide treatment for thrombocythemia in chronic myeloproliferative disorders. Haematologica. 2004;89:520–527. - PubMed
-
- Petrides PE, Gisslinger H, Steurer M, et al. Pharmacokinetics, bioequivalence, tolerability, and effects on platelet counts of two formulations of anagrelide in healthy volunteers and patients with thrombocythemia associated with chronic myeloproliferation. Clin Ther. 2009;31:386–398. doi: 10.1016/j.clinthera.2009.02.008. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

