CK2-mediated hyperphosphorylation of topoisomerase I targets serine 506, enhances topoisomerase I-DNA binding, and increases cellular camptothecin sensitivity
- PMID: 23185622
- PMCID: PMC3503890
- DOI: 10.1371/journal.pone.0050427
CK2-mediated hyperphosphorylation of topoisomerase I targets serine 506, enhances topoisomerase I-DNA binding, and increases cellular camptothecin sensitivity
Abstract
Topoisomerase I is the target for a potent class of chemotherapeutic drugs derived from the plant alkaloid camptothecin that includes irinotecan and topotecan. In this study we have identified a novel site of CK2-mediated topoisomerase I (topo I) phosphorylation at serine 506 (PS506) that is relevant to topo I function and to cellular responses to these topo I-targeted drugs. CK2 treatment induced hyperphosphorylation of recombinant topo I and expression of the PS506 epitope, and resulted in increased binding of topo I to supercoiled plasmid DNA. Hyperphosphorylated topo I was approximately three times more effective than the basal phosphorylated enzyme at relaxing plasmid supercoils but had similar DNA cleavage activity once bound to DNA. The PS506 epitope was expressed in cancer cell lines with elevated CK2 activity, hyperphosphorylated topo I, and increased sensitivity to camptothecin. In contrast, PS506 was not detected in normal cells or cancer cell lines with lower levels of CK2 activity. By experimentally manipulating CK2 activity in cancer cell lines, we demonstrate a cause and effect relationship between CK2 activity, PS506 expression, camptothecin-induced cellular DNA damage, and cellular camptothecin sensitivity. Our results show that the PS506 epitope is an indicator of dysregulated, hyperphosphorylated topo I in cancer cells, and may thus serve as a diagnostic or prognostic biomarker and predict tumor responsiveness to widely used topo I-targeted therapies.
Conflict of interest statement
Figures
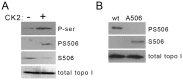

 ) followed by catalytic single-strand nicking (intermediate
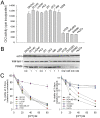
) followed by catalytic single-strand nicking (intermediate  ). (D) Time course of non-covalent association of 0.3 pmol basal (•) or hyperphosphorylated (○) R-topo I to 0.03 pmol of radiolabeled plasmid DNA. Topo I–DNA complexes were recovered and DNA quantified by scintillation counting. Results show the % of input DNA present in DNA–topo I complexes. (E) Catalytic rate of basal (•) and hyperphosphorylated (○) R-topo I on a synthetic suicide substrate following the formation of non-covalent complexes at 4°C. (F) Diagram of hairpin structure of suicide substrate showing topo I cleavage sequence (↓) and blocked 5′-end carrying a phosphate group (•). Diagram adapted from reference with permission from Oxford University Press.
). (D) Time course of non-covalent association of 0.3 pmol basal (•) or hyperphosphorylated (○) R-topo I to 0.03 pmol of radiolabeled plasmid DNA. Topo I–DNA complexes were recovered and DNA quantified by scintillation counting. Results show the % of input DNA present in DNA–topo I complexes. (E) Catalytic rate of basal (•) and hyperphosphorylated (○) R-topo I on a synthetic suicide substrate following the formation of non-covalent complexes at 4°C. (F) Diagram of hairpin structure of suicide substrate showing topo I cleavage sequence (↓) and blocked 5′-end carrying a phosphate group (•). Diagram adapted from reference with permission from Oxford University Press.
References
-
- Pommier Y, Pourquier P, Fan Y, Strumberg D (1998) Mechanism of action of eukaryotic DNA topoisomerase I and drugs targeted to the enzyme. Biochim Biophys Acta 1400: 83–105. - PubMed
-
- Wang JC (1996) DNA topoisomerases. Annu Rev Biochem 65: 635–692. - PubMed
-
- Champoux JJ (2001) DNA topoisomerases: structure, function, and mechanism. Annu Rev Biochem 70: 369–413. - PubMed
-
- Tsao YP, Russo A, Nyamuswa G, Silber R, Liu LF (1993) Interaction between replication forks and topoisomerase I-DNA cleavable complexes: studies in a cell-free SV40 DNA replication system. Cancer Res 53: 5908–5914. - PubMed
-
- Liu LF (1989) DNA topoisomerase poisons as antitumor drugs. Annu Rev Biochem 58: 351–375. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

