Investigation of follicular and non-follicular pathways for polyarginine and oleic acid-modified nanoparticles
- PMID: 23187866
- PMCID: PMC3760706
- DOI: 10.1007/s11095-012-0939-6
Investigation of follicular and non-follicular pathways for polyarginine and oleic acid-modified nanoparticles
Abstract
Purpose: To investigate the percutaneous permeation pathways of cell penetrating peptide modified lipid nanoparticles and oleic acid modified polymeric nanoparticles.
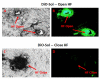
Methods: Confocal microscopy was performed on skin cultures (EpiDermFT™) for modified and un-modified nanoparticles. Differential stripping was performed following in vitro skin permeation of Ibuprofen (Ibu) encapsulated nanoparticles to estimate Ibu levels in different skin layers and receiver compartment. The hair follicles (HF) were blocked and in vitro skin permeation of nanoparticles was then compared with unblocked HF. The surface modified nanoparticles were investigated for response on allergic contact dermatitis (ACD).
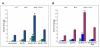
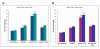
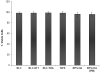
Results: Surface modified nanoparticles showed a significant higher (p < 0.05) in fluorescence in EpiDermFT™ cultures compared to controls. The HF play less than 5% role in total nanoparticle permeation into the skin. The Ibu levels were significantly high (p < 0.05) for surface modified nanoparticles compared to controls. The Ibu levels in skin and receiver compartment were not significantly different when HF were open or closed. Modified nanoparticles showed significant improvement in treatment of ACD compared to solution.
Conclusions: Our studies demonstrate that increased skin permeation of surface modified nanoparticles is not only dependent on a follicular pathway but also occur through non-follicular pathway(s).
Figures





Similar articles
-
Enhanced skin permeation using polyarginine modified nanostructured lipid carriers.J Control Release. 2012 Aug 10;161(3):735-45. doi: 10.1016/j.jconrel.2012.05.011. Epub 2012 May 14. J Control Release. 2012. PMID: 22617521 Free PMC article.
-
Effect of oleic acid modified polymeric bilayered nanoparticles on percutaneous delivery of spantide II and ketoprofen.J Control Release. 2012 Mar 10;158(2):336-45. doi: 10.1016/j.jconrel.2011.11.016. Epub 2011 Nov 21. J Control Release. 2012. PMID: 22134117 Free PMC article.
-
Transdermal self-permeation enhancement of ibuprofen.J Control Release. 2004 Nov 24;100(2):199-209. doi: 10.1016/j.jconrel.2004.08.011. J Control Release. 2004. PMID: 15544868
-
Delivery of ibuprofen to the skin.Int J Pharm. 2013 Nov 30;457(1):9-13. doi: 10.1016/j.ijpharm.2013.09.019. Epub 2013 Sep 21. Int J Pharm. 2013. PMID: 24064201 Review.
-
Applications and limitations of lipid nanoparticles in dermal and transdermal drug delivery via the follicular route.Eur J Pharm Biopharm. 2015 Nov;97(Pt A):152-63. doi: 10.1016/j.ejpb.2015.06.020. Epub 2015 Jul 2. Eur J Pharm Biopharm. 2015. PMID: 26144664 Review.
Cited by
-
Formulation Development and Investigations on Therapeutic Potential of Nanogel from Beta vulgaris L. Extract in Testosterone-Induced Alopecia.Biomed Res Int. 2023 Jan 31;2023:1777631. doi: 10.1155/2023/1777631. eCollection 2023. Biomed Res Int. 2023. PMID: 36760474 Free PMC article.
-
pH-Responsive Polypeptide-Based Smart Nano-Carriers for Theranostic Applications.Molecules. 2019 Aug 15;24(16):2961. doi: 10.3390/molecules24162961. Molecules. 2019. PMID: 31443287 Free PMC article. Review.
-
Current application of phytocompound-based nanocosmeceuticals for beauty and skin therapy.Int J Nanomedicine. 2016 May 11;11:1987-2007. doi: 10.2147/IJN.S104701. eCollection 2016. Int J Nanomedicine. 2016. PMID: 27274231 Free PMC article. Review.
-
Enhancing Permeation of Drug Molecules Across the Skin via Delivery in Nanocarriers: Novel Strategies for Effective Transdermal Applications.Front Bioeng Biotechnol. 2021 Mar 29;9:646554. doi: 10.3389/fbioe.2021.646554. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 33855015 Free PMC article. Review.
-
Emulgel: A Cutting Edge Approach for Topical Drug Delivery System.Curr Drug Res Rev. 2025;17(2):217-236. doi: 10.2174/0125899775278612240129055753. Curr Drug Res Rev. 2025. PMID: 38303537 Review.
References
-
- Meidan VM, Bonner MC, Michniak BB. Transfollicular drug delivery--is it a reality? Int J Pharm. 2005;306(1-2):1–14. - PubMed
-
- Lademann J, Richter H, Meinke M, Sterry W, Patzelt A. Which skin model is the most appropriate for the investigation of topically applied substances into the hair follicles? Skin Pharmacol Physiol. 2010;23(1):47–52. - PubMed
-
- Mehnert W, Mader K. Solid lipid nanoparticles: production, characterization and applications. Adv Drug Deliv Rev. 2001;47(2-3):165–196. - PubMed
-
- Mundargi RC, Babu VR, Rangaswamy V, Patel P, Aminabhavi TM. Nano/micro technologies for delivering macromolecular therapeutics using poly(D,L-lactide-co-glycolide) and its derivatives. J Control Release. 2008;125(3):193–209. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

