Cloning, expression, purification, crystallization and preliminary X-ray analysis of a fructokinase from Vibrio cholerae O395
- PMID: 23192049
- PMCID: PMC3509990
- DOI: 10.1107/S1744309112047598
Cloning, expression, purification, crystallization and preliminary X-ray analysis of a fructokinase from Vibrio cholerae O395
Abstract
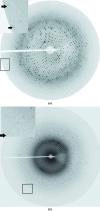
Fructokinase (FK), one of the crucial enzymes for sugar metabolism in bacterial systems, catalyses the unidirectional phosphorylation reaction from fructose to fructose 6-phosphate, thereby allowing parallel entry of fructose into glycolysis beside glucose. The cscK gene from Vibrio cholerae O395 coding for the enzyme FK has been cloned, overexpressed in Escherichia coli BL21 (DE3) and purified using Ni-NTA affinity chromatography. Crystals of V. cholerae FK (Vc-FK) and its cocrystal with fructose, adenosine diphosphate (ADP) and Mg2+ were grown in the presence of polyethylene glycol 6000 and diffracted to 2.45 and 1.75 Å resolution, respectively. Analysis of the diffraction data showed that both crystal forms have symmetry consistent with space group P2(1)2(1)2, but with different unit-cell parameters. Assuming the presence of two molecules in the asymmetric unit, the Matthews coefficient for the apo Vc-FK crystals was estimated to be 2.4 Å3 Da(-1), which corresponds to a solvent content of 48%. The corresponding values for the ADP- and sugar-bound Vc-FK crystals were 2.1 Å3 Da(-1) and 40%, respectively, assuming the presence of one molecule in the asymmetric unit.
Figures


Similar articles
-
Crystallization and preliminary X-ray analysis of a ribokinase from Vibrio cholerae O395.Acta Crystallogr F Struct Biol Commun. 2014 Aug;70(Pt 8):1098-102. doi: 10.1107/S2053230X14014411. Epub 2014 Jul 23. Acta Crystallogr F Struct Biol Commun. 2014. PMID: 25084391 Free PMC article.
-
Cloning, expression, purification, crystallization and preliminary X-ray analysis of the 31 kDa Vibrio cholerae heat-shock protein VcHsp31.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2011 Nov 1;67(Pt 11):1382-5. doi: 10.1107/S1744309111032970. Epub 2011 Oct 27. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2011. PMID: 22102237 Free PMC article.
-
Cloning, purification, crystallization and preliminary X-ray analysis of two low-molecular-weight protein tyrosine phosphatases from Vibrio cholerae.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012 Oct 1;68(Pt 10):1204-8. doi: 10.1107/S174430911203518X. Epub 2012 Sep 26. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012. PMID: 23027748 Free PMC article.
-
Overexpression, purification, crystallization and preliminary X-ray analysis of CheY4 from Vibrio cholerae O395.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2011 Dec 1;67(Pt 12):1645-8. doi: 10.1107/S1744309111041972. Epub 2011 Nov 30. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2011. PMID: 22139188 Free PMC article.
-
Cloning, overexpression, purification, crystallization and preliminary X-ray analysis of CheY3, a response regulator that directly interacts with the flagellar 'switch complex' in Vibrio cholerae.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2010 Aug 1;66(Pt 8):944-7. doi: 10.1107/S1744309110022797. Epub 2010 Jul 29. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2010. PMID: 20693676 Free PMC article.
Cited by
-
Large-scale conformational changes and redistribution of surface negative charge upon sugar binding dictate the fidelity of phosphorylation in Vibrio cholerae fructokinase.Sci Rep. 2018 Nov 16;8(1):16925. doi: 10.1038/s41598-018-35236-3. Sci Rep. 2018. PMID: 30446722 Free PMC article.
References
-
- Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. (1990). J. Mol. Biol. 215, 403–410. - PubMed
-
- Brünger, A. T., Adams, P. D., Clore, G. M., DeLano, W. L., Gros, P., Grosse-Kunstleve, R. W., Jiang, J.-S., Kuszewski, J., Nilges, M., Pannu, N. S., Read, R. J., Rice, L. M., Simonson, T. & Warren, G. L. (1998). Acta Cryst. D54, 905–921. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

