Cell therapy with human renal cell cultures containing erythropoietin-positive cells improves chronic kidney injury
- PMID: 23197816
- PMCID: PMC3659702
- DOI: 10.5966/sctm.2011-0048
Cell therapy with human renal cell cultures containing erythropoietin-positive cells improves chronic kidney injury
Abstract
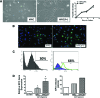
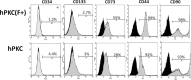
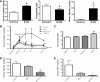
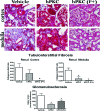
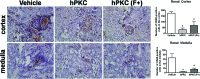
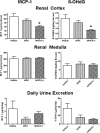
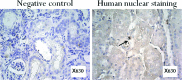
New therapeutic strategies for chronic kidney disease (CKD) are necessary to offset the rising incidence of CKD and donor shortage. Erythropoietin (EPO), a cytokine produced by fibroblast-like cells in the kidney, has recently emerged as a renoprotective factor with anti-inflammatory, antioxidant properties. This study (a) determined whether human renal cultures (human primary kidney cells [hPKC]) can be enriched in EPO-positive cells (hPKC(F+)) by using magnetic-bead sorting; (b) characterized hPKC(F+) following cell separation; and (c) established that intrarenal delivery of enriched hPKC(F+) cells would be more beneficial in treatment of renal injury, inflammation, and oxidative stress than unsorted hPKC cultures in a chronic kidney injury model. Fluorescence-activated cell sorting analysis revealed higher expression of EPO (36%) and CD73 (27%) in hPKC(F+) as compared with hPKC. After induction of renal injury, intrarenal delivery of hPKC(F+) or hPKC significantly reduced serum creatinine, interstitial fibrosis in the medulla, and abundance of CD68-positive cells in the cortex and medulla (p < .05). However, only hPKC(F+) attenuated interstitial fibrosis in the renal cortex and decreased urinary albumin (3.5-fold) and urinary tubular injury marker kidney injury molecule 1 (16-fold). hPKC(F+) also significantly reduced levels of renal cortical monocyte chemotactic protein 1 (1.8-fold) and oxidative DNA marker 8-hydroxy-deoxyguanosine (8-OHdG) (2.4-fold). After 12 weeks, we detected few injected cells, which were localized mostly to the cortical interstitium. Although cell therapy with either hPKC(F+) or hPKC improved renal function, the hPKC(F+) subpopulation provides greater renoprotection, perhaps through attenuation of inflammation and oxidative stress. We conclude that hPKC(F+) may be used as components of cell-based therapies for degenerative kidney diseases.
Figures
References
-
- Al-Awqati Q, Oliver JA. Stem cells in the kidney. Kidney Int. 2002;61:387–395. - PubMed
-
- Benigni A, Morigi M, Remuzzi G. Kidney regeneration. Lancet. 2010;375:1310–1317. - PubMed
-
- Guo JK, Cantley LG. Cellular maintenance and repair of the kidney. Annu Rev Physiol. 2010;72:357–376. - PubMed
-
- U.S. Renal Data System. Minneapolis, MN: U.S. Renal Data System Coordinating Center; 2009. U.S. Renal Data System Annual Report.
-
- Zenovich AG, Taylor DA. Cell therapy in kidney disease: Cautious optimism … but optimism nonetheless. Perit Dial Int. 2007;27(suppl 2):S94–S103. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

