Variability in the generation of induced pluripotent stem cells: importance for disease modeling
- PMID: 23197870
- PMCID: PMC3659735
- DOI: 10.5966/sctm.2012-0043
Variability in the generation of induced pluripotent stem cells: importance for disease modeling
Abstract
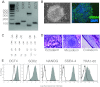
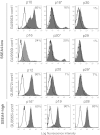
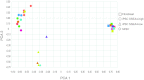
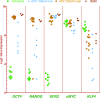
In the field of disease modeling, induced pluripotent stem cells (iPSCs) have become an appealing choice, especially for diseases that do not have an animal model. They can be generated from patients with known clinical features and compared with cells from healthy controls to identify the biological bases of disease. This study was undertaken to determine the variability in iPSC lines derived from different individuals, with the aim of determining criteria for selecting iPSC lines for disease models. We generated and characterized 18 iPSC lines from eight donors and considered variability at three levels: (a) variability in the criteria that define iPSC lines as pluripotent cells, (b) variability in cell lines from different donors, and (c) variability in cell lines from the same donor. We found that variability in transgene expression and pluripotency marker levels did not prevent iPSCs from fulfilling all other criteria for pluripotency, including teratoma formation. We found low interindividual and interclonal variability in iPSCs that fulfilled the most stringent criteria for pluripotency, with very high correlation in their gene expression profiles. Interestingly, some cell lines exhibited reprogramming instability, spontaneously regressing from a fully to a partially reprogrammed state. This was associated with a low percentage of cells expressing the pluripotency marker stage-specific embryonic antigen-4. Our study shows that it is possible to define a similar "ground state" for each cell line as the basis for making patient versus control comparisons, an essential step in order to identify disease-associated variability above individual and cell line variability.
Figures

References
-
- Vitale AM, Wolvetang E, Mackay-Sim A. Induced pluripotent stem cells: A new technology to study human diseases. Int J Biochem Cell Biol. 2011;43:843–846. - PubMed
-
- Frumkin T, Malcov M, Telias M, et al. Human embryonic stem cells carrying mutations for severe genetic disorders. In Vitro Cell Dev Biol Anim. 2010;46:327–336. - PubMed
-
- Pickering SJ, Minger SL, Patel M, et al. Generation of a human embryonic stem cell line encoding the cystic fibrosis mutation deltaF508, using preimplantation genetic diagnosis. Reprod Biomed Online. 2005;10:390–397. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

