Robot-assisted minimally invasive thoraco-laparoscopic esophagectomy versus open transthoracic esophagectomy for resectable esophageal cancer, a randomized controlled trial (ROBOT trial)
- PMID: 23199187
- PMCID: PMC3564860
- DOI: 10.1186/1745-6215-13-230
Robot-assisted minimally invasive thoraco-laparoscopic esophagectomy versus open transthoracic esophagectomy for resectable esophageal cancer, a randomized controlled trial (ROBOT trial)
Abstract
Background: For esophageal cancer patients, radical esophagolymphadenectomy is the cornerstone of multimodality treatment with curative intent. Transthoracic esophagectomy is the preferred surgical approach worldwide allowing for en-bloc resection of the tumor with the surrounding lymph nodes. However, the percentage of cardiopulmonary complications associated with the transthoracic approach is high (50 to 70%).Recent studies have shown that robot-assisted minimally invasive thoraco-laparoscopic esophagectomy (RATE) is at least equivalent to the open transthoracic approach for esophageal cancer in terms of short-term oncological outcomes. RATE was accompanied with reduced blood loss, shorter ICU stay and improved lymph node retrieval compared with open esophagectomy, and the pulmonary complication rate, hospital stay and perioperative mortality were comparable. The objective is to evaluate the efficacy, risks, quality of life and cost-effectiveness of RATE as an alternative to open transthoracic esophagectomy for treatment of esophageal cancer.
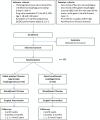
Methods/design: This is an investigator-initiated and investigator-driven monocenter randomized controlled parallel-group, superiority trial. All adult patients (age ≥ 18 and ≤ 80 years) with histologically proven, surgically resectable (cT1-4a, N0-3, M0) esophageal carcinoma of the intrathoracic esophagus and with European Clinical Oncology Group performance status 0, 1 or 2 will be assessed for eligibility and included after obtaining informed consent. Patients (n = 112) with resectable esophageal cancer are randomized in the outpatient department to either RATE (n = 56) or open three-stage transthoracic esophageal resection (n = 56). The primary outcome of this study is the percentage of overall complications (grade 2 and higher) as stated by the modified Clavien-Dindo classification of surgical complications.
Discussion: This is the first randomized controlled trial designed to compare RATE with open transthoracic esophagectomy as surgical treatment for resectable esophageal cancer. If our hypothesis is proven correct, RATE will result in a lower percentage of postoperative complications, lower blood loss, and shorter hospital stay, but with at least similar oncologic outcomes and better postoperative quality of life compared with open transthoracic esophagectomy. The study started in January 2012. Follow-up will be 5 years. Short-term results will be analyzed and published after discharge of the last randomized patient.
Trial registration: Dutch trial register: NTR3291 ClinicalTrial.gov: NCT01544790.
Figures

References
-
- Omloo JMT, Lagarde SM, Hulscher JBF, Reitsma JB, Fockens P, van Dekken H, ten Kate FJW, Obertop H, Tilanus HW, Lanschot JJB. Extended transthoracic resection compared with limited transhiatal resection for adenocarcinoma of the mid/distal esophagus. Ann Surg. 2007;246:992–1001. doi: 10.1097/SLA.0b013e31815c4037. - DOI - PubMed
-
- Burmeister BH, Smithers BM, Gebski V, Fitzgerald L, Simes RJ, Devitt P, Ackland S, Gotley DC, Joseph D, Millar J, North J, Walpole ET, Denham JW. Trans-Tasman Radiation Oncology Group; Australasian Gastro-Intestinal Trials Group. Surgery alone versus chemoradiotherapy followed by surgery for resectable cancer of the oesophagus: a randomised controlled phase III trial. Lancet Oncol. 2005;6:659–668. doi: 10.1016/S1470-2045(05)70288-6. - DOI - PubMed
-
- Gebski V, Burmeister B, Smithers BM, Foo K, Zalcberg J, Simes J. Australasian Gastro-Intestinal Trials Group. Survival benefits from neoadjuvant chemoradiotherapy or chemotherapy in oesophageal carcinoma: a meta-analysis. Lancet Oncol. 2007;8:226–234. doi: 10.1016/S1470-2045(07)70039-6. - DOI - PubMed
Publication types
MeSH terms
Supplementary concepts
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials

