Protein Arginine Methyltransferases (PRMTs): promising targets for the treatment of pulmonary disorders
- PMID: 23202904
- PMCID: PMC3497278
- DOI: 10.3390/ijms131012383
Protein Arginine Methyltransferases (PRMTs): promising targets for the treatment of pulmonary disorders
Abstract
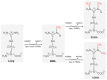
Protein arginine methylation is a novel posttranslational modification that plays a pivotal role in a variety of intracellular events, such as signal transduction, protein-protein interaction and transcriptional regulation, either by the direct regulation of protein function or by metabolic products originating from protein arginine methylation that influence nitric oxide (NO)-dependent processes. A growing body of evidence suggests that both mechanisms are implicated in cardiovascular and pulmonary diseases. This review will present and discuss recent research on PRMTs and the methylation of non-histone proteins and its consequences for the pathogenesis of various lung disorders, including lung cancer, pulmonary fibrosis, pulmonary hypertension, chronic obstructive pulmonary disease and asthma. This article will also highlight novel directions for possible future investigations to evaluate the functional contribution of arginine methylation in lung homeostasis and disease.
Figures

References
-
- Heron M., Hoyert D.L., Murphy S.L., Xu J., Kochanek K.D., Tejada-Vera B. Deaths: Final data for 2006. Natl. Vital Stat. Rep. 2009;57:1–134. - PubMed
-
- Yach D., Hawkes C., Gould C.L., Hofman K.J. The global burden of chronic diseases: Overcoming impediments to prevention and control. JAMA J. Am. Med. Assoc. 2004;291:2616–2622. - PubMed
-
- Orens J.B., Shearon T.H., Freudenberger R.S., Conte J.V., Bhorade S.M., Ardehali A. Thoracic organ transplantation in the United States, 1995–2004. Am. J. Transplant. 2006;6:1188–1197. - PubMed
-
- Trulock E.P., Christie J.D., Edwards L.B., Boucek M.M., Aurora P., Taylor D.O., Dobbels F., Rahmel A.O., Keck B.M., Hertz M.I. Registry of the international society for heart and lung transplantation: Twenty-fourth official adult lung and heart-lung transplantation report—2007. J. Heart Lung Transplant. 2007;26:782–795. - PubMed
-
- O’Beirne S., Counihan I.P., Keane M.P. Interstitial lung disease and lung transplantation. Semin. Respir. Crit. Care Med. 2010;31:139–146. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

