Structural mechanism of ER retrieval of MHC class I by cowpox
- PMID: 23209377
- PMCID: PMC3507924
- DOI: 10.1371/journal.pbio.1001432
Structural mechanism of ER retrieval of MHC class I by cowpox
Abstract
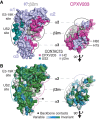
One of the hallmarks of viral immune evasion is the capacity to disrupt major histocompatibility complex class I (MHCI) antigen presentation to evade T-cell detection. Cowpox virus encoded protein CPXV203 blocks MHCI surface expression by exploiting the KDEL-receptor recycling pathway, and here we show that CPXV203 directly binds a wide array of fully assembled MHCI proteins, both classical and non-classical. Further, the stability of CPXV203/MHCI complexes is highly pH dependent, with dramatically increased affinities at the lower pH of the Golgi relative to the endoplasmic reticulum (ER). Crystallographic studies reveal that CPXV203 adopts a beta-sandwich fold similar to poxvirus chemokine binding proteins, and binds the same highly conserved MHCI determinants located under the peptide-binding platform that tapasin, CD8, and natural killer (NK)-receptors engage. Mutagenesis of the CPXV203/MHCI interface identified the importance of two CPXV203 His residues that confer low pH stabilization of the complex and are critical to ER retrieval of MHCI. These studies clarify mechanistically how CPXV203 coordinates with other cowpox proteins to thwart antigen presentation.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures





Comment in
-
How cowpox virus turns back cell defenses.PLoS Biol. 2012;10(11):e1001431. doi: 10.1371/journal.pbio.1001431. Epub 2012 Nov 27. PLoS Biol. 2012. PMID: 23209376 Free PMC article. No abstract available.
Similar articles
-
Cowpox virus employs a two-pronged strategy to outflank MHCI antigen presentation.Mol Immunol. 2013 Sep;55(2):156-8. doi: 10.1016/j.molimm.2012.11.011. Epub 2013 Jan 10. Mol Immunol. 2013. PMID: 23312338 Free PMC article. Review.
-
Two mechanistically distinct immune evasion proteins of cowpox virus combine to avoid antiviral CD8 T cells.Cell Host Microbe. 2009 Nov 19;6(5):422-32. doi: 10.1016/j.chom.2009.09.012. Cell Host Microbe. 2009. PMID: 19917497 Free PMC article.
-
Cowpox virus exploits the endoplasmic reticulum retention pathway to inhibit MHC class I transport to the cell surface.Cell Host Microbe. 2007 Nov 15;2(5):306-15. doi: 10.1016/j.chom.2007.09.002. Cell Host Microbe. 2007. PMID: 18005752
-
Cowpox virus inhibits the transporter associated with antigen processing to evade T cell recognition.Cell Host Microbe. 2009 Nov 19;6(5):433-45. doi: 10.1016/j.chom.2009.09.013. Cell Host Microbe. 2009. PMID: 19917498 Free PMC article.
-
Modulation of the host immune response by cowpox virus.Microbes Infect. 2010 Nov;12(12-13):900-9. doi: 10.1016/j.micinf.2010.07.007. Epub 2010 Jul 29. Microbes Infect. 2010. PMID: 20673807 Free PMC article. Review.
Cited by
-
KDEL Receptors: Pathophysiological Functions, Therapeutic Options, and Biotechnological Opportunities.Biomedicines. 2022 May 25;10(6):1234. doi: 10.3390/biomedicines10061234. Biomedicines. 2022. PMID: 35740256 Free PMC article. Review.
-
Structural Models for Roseolovirus U20 And U21: Non-Classical MHC-I Like Proteins From HHV-6A, HHV-6B, and HHV-7.Front Immunol. 2022 Apr 4;13:864898. doi: 10.3389/fimmu.2022.864898. eCollection 2022. Front Immunol. 2022. PMID: 35444636 Free PMC article. Review.
-
A negative feedback modulator of antigen processing evolved from a frameshift in the cowpox virus genome.PLoS Pathog. 2014 Dec 11;10(12):e1004554. doi: 10.1371/journal.ppat.1004554. eCollection 2014 Dec. PLoS Pathog. 2014. PMID: 25503639 Free PMC article.
-
Structural Conservation and Functional Diversity of the Poxvirus Immune Evasion (PIE) Domain Superfamily.Viruses. 2015 Aug 28;7(9):4878-98. doi: 10.3390/v7092848. Viruses. 2015. PMID: 26343707 Free PMC article. Review.
-
Direct Priming of CD8+ T Cells Persists in the Face of Cowpox Virus Inhibitors of Antigen Presentation.J Virol. 2021 Apr 26;95(10):e00186-21. doi: 10.1128/JVI.00186-21. Epub 2021 Mar 10. J Virol. 2021. PMID: 33692206 Free PMC article.
References
-
- Hansen TH, Bouvier M (2009) MHC class I antigen presentation: learning from viral evasion strategies. Nat Rev Immunol 9: 503–513. - PubMed
-
- Lilley BN, Ploegh HL (2005) Viral modulation of antigen presentation: manipulation of cellular targets in the ER and beyond. Immunol Rev 207: 126–144. - PubMed
-
- Früh K, Ahn K, Djaballah H, Sempé P, van Endert PM, et al. (1995) A viral inhibitor of peptide transporters for antigen presentation. Nature 375: 415–418. - PubMed
-
- Hill A, Jugovic P, York I, Russ G, Bennink J, et al. (1995) Herpes simplex virus turns off the TAP to evade host immunity. Nature 375: 411–415. - PubMed
-
- Ahn K, Gruhler A, Galocha B, Jones TR, Wiertz EJ, et al. (1997) The ER-luminal domain of the HCMV glycoprotein US6 inhibits peptide translocation by TAP. Immunity 6: 613–621. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

