Trypanosoma cruzi SSP4 Amastigote Protein Induces Expression of Immunoregulatory and Immunosuppressive Molecules in Peripheral Blood Mononuclear Cells
- PMID: 23209478
- PMCID: PMC3503440
- DOI: 10.1155/2012/829139
Trypanosoma cruzi SSP4 Amastigote Protein Induces Expression of Immunoregulatory and Immunosuppressive Molecules in Peripheral Blood Mononuclear Cells
Abstract
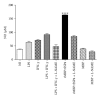
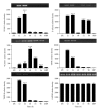
The acute phase of Chagas' disease in mice and human is marked by states of immunosuppression, in which Trypanosoma cruzi replicates extensively and releases immunomodulatory molecules that delay parasite-specific responses mediated by effector T cells. This mechanism of evasion allows the parasite to spread in the host. Parasite molecules that regulate the host immune response during Chagas' disease have not been fully identified, particularly proteins of the amastigote stage. In this work, we evaluated the role of the GPI anchored SSP4 protein of T. cruzi as an immunomodulatory molecule in peripheral blood mononuclear cells (PBMCs). rMBP::SSP4 protein was able to stimulate nitric oxide (NO) production. Likewise, rMBP::SSP4 induced the expression of genes and production of molecules involved in the inflammatory process, such as, cytokines, chemokines, and adhesion molecules (CAMs) as determined by RT-PCR and ELISA. These results suggest that the amastigote SSP4 molecule could play a key role in the immunoregulatory and/or immunosuppressive process observed in the acute phase of infection with T. cruzi.
Figures


References
-
- Schofield CJ, Jannin J, Salvatella R. The future of Chagas disease control. Trends in Parasitology. 2006;22(12):583–588. - PubMed
-
- Tanowitz HB, Gumprecht JP, Spurr D, et al. Cytokine gene expression of endothelial cells infected with Trypanosoma cruzi . Journal of Infectious Diseases. 1992;166(3):598–603. - PubMed
-
- Teixeira MM, Gazzinelli RT, Silva JS. Chemokines, inflammation and Trypanosoma cruzi infection. Trends in Parasitology. 2002;18(6):262–265. - PubMed
LinkOut - more resources
Full Text Sources

